Toxin-antitoxin systems
Toxin-antitoxin (TA) systems are small genetic elements found on plasmids or chromosomes of many bacteria and Archaea. Under normal growth conditions, the activity of the toxin protein or its translation is counteracted by an antitoxin protein or a non-coding antitoxin sRNA (see below for classification of TA types). Toxins are stable whereas antitoxins are metabolically unstable so that, unless the antitoxin is continuously expressed, the free toxin accumulates and exerts its toxic effect  .
.
Changes in the physiological conditions, like stress conditions or viral infection can result in the degradation of the antitoxin which is highly unstable. Proteases (e.g. Lon, Clp, Hlfp) have be linked to the degradation of many antitoxins. Unleashed toxin proteins impede or alter cellular processes including translation, cell division, DNA replication, ATP synthesis, mRNA stability or cell wall synthesis and leads to dormancy. This dormant state probably enables the bacteria to survive environmentally unfavorable conditions but may also sometimes lead to cell death 

 .
.
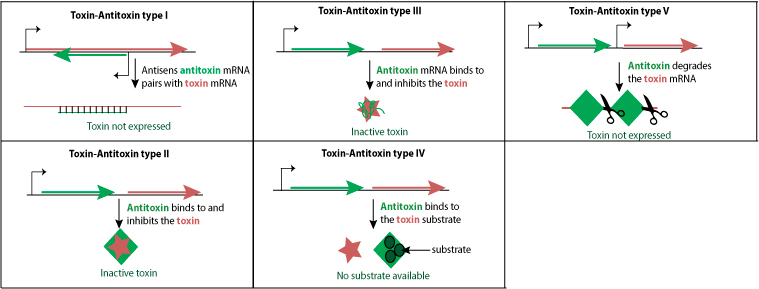
The modularity of the TA systems whereby toxins and antitoxins can have more than one partner gives rise to a network of complex, interconnected families of TA systems.
The number of TA systems is variable: E.coli K12 codes for least 33 TA systems, M.tuberculosis than 60 whereas M.segmatis has only two so far.
Toxin-antitoxin systems as antiviral defense
Some TA systems act as antiviral factors: if the infecting virus inhibits the host cell's transcription and/or translation, which is often the case, the unstable antitoxin disappears and the toxin exerts its toxic effect to limit viral replication. The majority of toxins in TA systems used as antiviral factors interfere with the translation process, mostly via mRNA or tRNA cleavage.
Prokaryotic viruses can bypass this defense system by expressing a product similar to the antitoxin and thereby inhibit host translation shutoff 

 .
.
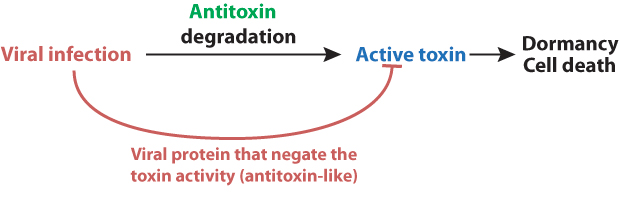
Here are some bacterial TA systems used as antiviral defense and the counteracting viral response:
| Host bacteria | Bacterial toxin | Bacterial antitoxin | Toxin activity | Family | Genus | Virus | strategy of evasion | ref. |
| Pectobacterium atrosepticum | ToxN | ToxI | mRNAs site-specific endoribonuclease | Myoviridae | unclassified | ΦTE | Viral peudo-ToxI |  |
| E.coli | MazF | MazE | mRNAs site-specific endoribonuclease | Myoviridae | Punalikevirus | Enterobacteria phage P1 | ? | 
 |
| E.coli | RnlA | RnlB | mRNAs endoribonuclease | Myoviridae | T4likevirus | Enterobacteria phage T4 | Viral antitoxin Dmd |   |
| E.coli | LsoA | LsoB | mRNAs endoribonuclease | Myoviridae | T4likevirus | Enterobacteria phage T4 | Viral antitoxin Dmd |   |
| L.lactis | AbiQ | ? | mRNAs endoribonuclease | Siphoviridae | unclassified | Lactococcus phage P008 | gp38 mediated? |  |
Toxin-antitoxin systems and persistence
On plasmids, TA systems aid plasmid maintenance in the host population. The host cell cannot get rid of the plasmid expressing the TA system because the antitoxin will be rapidly degraded unlike the toxin which is much more stable. In the same way, TA systems can be exploited by viruses to favor the persistence of TA bearing prophages  .
.
Simon J Unterholzner, Brigitte Poppenberger, Wilfried Rozhon
Mob Genet Elements September 1, 2013; 3: e26219
Yoshihiro Yamaguchi, Jung-Ho Park, Masayori Inouye
Annu. Rev. Genet. 2011; 45: 61-79
Iwona Mruk, Ichizo Kobayashi
Nucleic Acids Res. January 2014; 42: 70-86
Hanna Engelberg-Kulka, Ronen Hazan, Shahar Amitai
J. Cell. Sci. October 1, 2005; 118: 4327-4332
D C Pecota, T K Wood
J. Bacteriol. April 1996; 178: 2044-2050
Tim R Blower, Terry J Evans, Rita Przybilski, Peter C Fineran, George P C Salmond
PLoS Genet. 2012; 8: e1003023
Kenta Naka, Mitsunori Koga, Tetsuro Yonesaki, Yuichi Otsuka
Mol. Microbiol. February 2014; 91: 596-605
Julie E Samson, Alfonso H Magad?n, Mourad Sabri, Sylvain Moineau
Nat. Rev. Microbiol. October 2013; 11: 675-687
H Lehnherr, E Maguin, S Jafri, M B Yarmolinsky
J. Mol. Biol. October 5, 1993; 233: 414-428
Oksana I Demidenok, Arseny S Kaprelyants, Anna V Goncharenko
FEMS Microbiol. Lett. March 2014; 352: 69-77
Kira S Makarova, Yuri I Wolf, Eugene V Koonin
Nucleic Acids Res. April 2013; 41: 4360-4377
Gregory M Cook, Jennifer R Robson, Rebekah A Frampton, Joanna McKenzie, Rita Przybilski, Peter C Fineran, Vickery L Arcus
Biochim. Biophys. Acta July 2013; 1829: 523-531
R Hazan, H Engelberg-Kulka
Mol. Genet. Genomics September 2004; 272: 227-234
Yong Wei, Zeng-Qiang Gao, Yuichi Otsuka, Kenta Naka, Tetsuro Yonesaki, Heng Zhang, Yu-Hui Dong
Mol. Microbiol. December 2013; 90: 956-965
Yuichi Otsuka, Tetsuro Yonesaki
Mol. Microbiol. February 2012; 83: 669-681
Yuichi Otsuka, Tetsuro Yonesaki
Mol. Microbiol. February 2012; 83: 669-681
Francesca L Short, Tim R Blower, George P C Salmond
Mol. Microbiol. February 2012; 83: 665-668
Julie E. Samson, Maxime B?langer, Sylvain Moineau
J. Bacteriol. September 2013; 195: 3947-3956
