Viral penetration into host nucleus (kw:KW-1163)
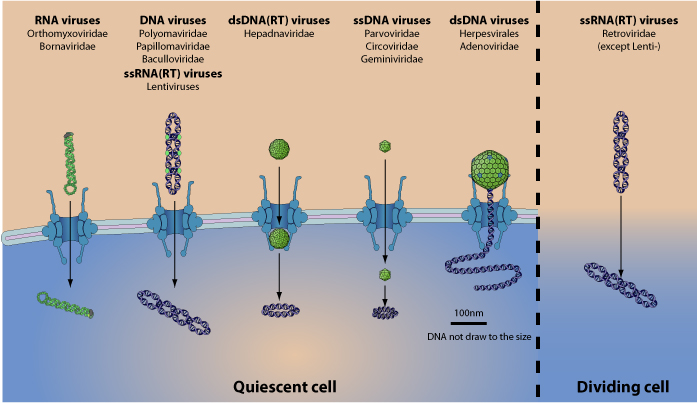
Most DNA and few RNA viruses target their genome to the host nucleus. The crossing of nuclear membrane occurs in several ways  :
:
-RNA virus, dsDNA virus and lentivirus genomes enter via the nuclear pore complex (NPC) through the cellular Importin transport.
-ssDNA virus capsid seems to be small enough to cross the NPC and enter the nucleus as an intact capsid.
-Hepadnaviridae capsid would enter the NPC pore, but remains attached to it and releases the viral genomic DNA into the nucleoplasm.
-Herpesvirales capsid is too large to enter the NPC pore, the viral genome is directly injected through the NPC on which the capsid docks.
-All retroviridae except lentivirus would enter the nucleus during mitosis, when the nuclear membrane temporarily disintegrates.
All these strategies to cross the nuclear envelope barrier are associated with various levels of capsid disassembly, since virus can pass intact (e.g. parvoviridae) or, in the case of injection, only the viral genome enters the nucleus (e.g. herpesviruses). Genome integration in the host genome may eventually follow
 .
.
Source: Viruses and the nuclear envelope,Thomas Hennig, Peter O'Hare, Curr. Opin. Cell Biol. June 2015; 34: 113.121
Thomas Hennig, Peter O'Hare
Curr. Opin. Cell Biol. June 2015; 34: 113-121
P. M. Ojala, B. Sodeik, M. W. Ebersold, U. Kutay, A. Helenius
Mol. Cell. Biol. July 2000; 20: 4922-4931
David Pasdeloup, Danielle Blondel, Anabela L. Isidro, Frazer J. Rixon
J. Virol. July 2009; 83: 6610-6623
Anna Maria Copeland, William W. Newcomb, Jay C. Brown
J. Virol. February 2009; 83: 1660?1668
Kathrin Rode, Katinka D?hner, Anne Binz, Mandy Glass, Tanja Strive, Rudolf Bauerfeind, Beate Sodeik
J. Virol. May 2011; 85: 4271?4283
J. Clever, M. Yamada, H. Kasamatsu
Proc. Natl. Acad. Sci. U.S.A. August 15, 1991; 88: 7333?7337
Veronika Butin-Israeli, Orly Ben-nun-Shaul, Idit Kopatz, Stephen A. Adam, Takeshi Shimi, Robert D. Goldman, Ariella Oppenheim
Nucleus August 2011; 2: 320?330
Sten Strunze, Lloyd C. Trotman, Karin Boucke, Urs F. Greber
Mol. Biol. Cell June 2005; 16: 2999?3009
L. C. Trotman, N. Mosberger, M. Fornerod, R. P. Stidwill, U. F. Greber
Nat. Cell Biol. December 2001; 3: 1092?1100
Sten Strunze, Martin F. Engelke, I.-Hsuan Wang, Daniel Puntener, Karin Boucke, Sibylle Schleich, Michael Way, Philipp Schoenenberger, Christoph J. Burckhardt, Urs F. Greber
Cell Host Microbe September 15, 2011; 10: 210?223
Clemence E. Hindley, Fiona J. Lawrence, David A. Matthews
Traffic October 2007; 8: 1313?1322
A. C. Saphire, T. Guan, E. C. Schirmer, G. R. Nemerow, L. Gerace
J. Biol. Chem. February 11, 2000; 275: 4298?4304
N. D. van Loo, E. Fortunati, E. Ehlert, M. Rabelink, F. Grosveld, B. J. Scholte
J. Virol. January 2001; 75: 961?970
Sarah Cohen, Igor Etingov, Nelly Pant?
Int Rev Cell Mol Biol 2012; 299: 117?159
J. Hansen, K. Qing, A. Srivastava
J. Virol. May 2001; 75: 4080?4090
Sarah C. Nicolson, R. Jude Samulski
J. Virol. April 2014; 88: 4132?4144
Manvi Porwal, Sarah Cohen, Kenza Snoussi, Ruth Popa-Wagner, Fenja Anderson, Nathalie Dugot-Senant, Harald Wodrich, Christiane Dinsart, J?rgen A. Kleinschmidt, Nelly Pant?, Michael Kann
PLoS Pathog. October 2013; 9: e1003671
Makoto Ozawa, Ken Fujii, Yukiko Muramoto, Shinya Yamada, Seiya Yamayoshi, Ayato Takada, Hideo Goto, Taisuke Horimoto, Yoshihiro Kawaoka
J. Virol. January 2007; 81: 30?41
R. E. O?Neill, R. Jaskunas, G. Blobel, P. Palese, J. Moroianu
J. Biol. Chem. September 29, 1995; 270: 22701?22704
Renate K?nig, Silke Stertz, Yingyao Zhou, Atsushi Inoue, H.-Heinrich Hoffmann, Suchita Bhattacharyya, Judith G. Alamares, Donna M. Tscherne, Mila B. Ortigoza, Yuhong Liang, Qinshan Gao, Shane E. Andrews, Sourav Bandyopadhyay, Paul De Jesus, Buu P. Tu, Lars Pache, Crystal Shih, Anthony Orth, Ghislain Bonamy, Loren Miraglia, Trey Ideker, Adolfo Garc?a-Sastre, John A. T. Young, Peter Palese, Megan L. Shaw, Sumit K. Chanda
Nature February 11, 2010; 463: 813?817
Alexander Karlas, Nikolaus Machuy, Yujin Shin, Klaus-Peter Pleissner, Anita Artarini, Dagmar Heuer, Daniel Becker, Hany Khalil, Lesley A. Ogilvie, Simone Hess, Andr? P. M?urer, Elke M?ller, Thorsten Wolff, Thomas Rudel, Thomas F. Meyer
Nature February 11, 2010; 463: 818?822
Tao Deng, Othmar G. Engelhardt, Benjamin Thomas, Alexandre V. Akoulitchev, George G. Brownlee, Ervin Fodor
J. Virol. December 2006; 80: 11911?11919
Sagi D. Shapira, Irit Gat-Viks, Bennett O. V. Shum, Amelie Dricot, Marciela M. de Grace, Liguo Wu, Piyush B. Gupta, Tong Hao, Serena J. Silver, David E. Root, David E. Hill, Aviv Regev, Nir Hacohen
Cell December 24, 2009; 139: 1255?1267
Guillaume Jacquot, Erwann Le Rouzic, Annie David, Julie Mazzolini, J?r?me Bouchet, Serge Bouaziz, Florence Niedergang, Gianfranco Pancino, Serge Benichou
Retrovirology 2007; 4: 84
Zhujun Ao, Kallesh Danappa Jayappa, Binchen Wang, Yingfeng Zheng, Xiaoxia Wang, Jinyu Peng, Xiaojian Yao
J. Biol. Chem. March 23, 2012; 287: 10544?10555
Lyubov Zaitseva, Peter Cherepanov, Lada Leyens, Sam J. Wilson, Jane Rasaiyaah, Ariberto Fassati
Retrovirology 2009; 6: 11
Hirotaka Ebina, Jun Aoki, Shunsuke Hatta, Takeshi Yoshida, Yoshio Koyanagi
Microbes Infect. July 2004; 6: 715?724
P. Gallay, T. Hope, D. Chin, D. Trono
Proc. Natl. Acad. Sci. U.S.A. September 2, 1997; 94: 9825?9830
Zhujun Ao, Kallesh Danappa Jayappa, Binchen Wang, Yingfeng Zheng, Sam Kung, Eric Rassart, Reinhard Depping, Matthias Kohler, Eric A. Cohen, Xiaojian Yao
J. Virol. September 2010; 84: 8650?8663
Lavanya Krishnan, Kenneth A. Matreyek, Ilker Oztop, Kyeongeun Lee, Christopher H. Tipper, Xiang Li, Mohd J. Dar, Vineet N. Kewalramani, Alan Engelman
J. Virol. January 2010; 84: 397?406
KyeongEun Lee, Zandrea Ambrose, Thomas D. Martin, Ilker Oztop, Alok Mulky, John G. Julias, Nick Vandegraaff, Joerg G. Baumann, Rui Wang, Wendy Yuen, Taichiro Takemura, Kenneth Shelton, Ichiro Taniuchi, Yuan Li, Joseph Sodroski, Dan R. Littman, John M. Coffin, Stephen H. Hughes, Derya Unutmaz, Alan Engelman, Vineet N. KewalRamani
Cell Host Microbe March 18, 2010; 7: 221?233
Torsten Schaller, Karen E. Ocwieja, Jane Rasaiyaah, Amanda J. Price, Troy L. Brady, Shoshannah L. Roth, St?phane Hu?, Adam J. Fletcher, KyeongEun Lee, Vineet N. KewalRamani, Mahdad Noursadeghi, Richard G. Jenner, Leo C. James, Frederic D. Bushman, Greg J. Towers
PLoS Pathog. December 2011; 7: e1002439
O. K. Haffar, S. Popov, L. Dubrovsky, I. Agostini, H. Tang, T. Pushkarsky, S. G. Nadler, M. Bukrinsky
J. Mol. Biol. June 2, 2000; 299: 359?368
Masahiro Yamashita, Michael Emerman
J. Virol. June 2004; 78: 5670?5678
Andr? Schmitz, Alexandra Schwarz, Michael Foss, Lixin Zhou, Birgit Rabe, Julia Hoellenriegel, Miriam Stoeber, Nelly Pant?, Michael Kann
PLoS Pathog. January 2010; 6: e1000741
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)598 entries grouped by strain
2 entries
Hepatitis delta virus genotype I (isolate D380) (HDV) reference strain
2 entries
Hepatitis delta virus genotype II (isolate TW2476) (HDV) reference strain
2 entries
Hepatitis delta virus genotype III (isolate VnzD8349) (HDV) reference strain
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate HXB2) (HIV-1) reference strain
2 entries
Human immunodeficiency virus type 1 group N (isolate YBF30) (HIV-1) reference strain
2 entries
Human immunodeficiency virus type 1 group O (isolate ANT70) (HIV-1) reference strain
2 entries
Human immunodeficiency virus type 2 subtype A (isolate BEN) (HIV-2) reference strain
1 entry
Abutilon mosaic virus (isolate West India) (AbMV) reference strain
1 entry
African cassava mosaic virus (isolate West Kenyan 844) (ACMV) (Cassava latent virus (isolate West Kenyan 844)) reference strain
1 entry
Alcelaphine herpesvirus 1 (strain C500) (AlHV-1) (Malignant catarrhal fever virus) reference strain
1 entry
BK polyomavirus (BKPyV) (Human polyomavirus 1) reference strain
1 entry
Bean golden yellow mosaic virus (isolate Puerto Rico) (BGYMV) (Bean golden mosaic virus (isolate Puerto Rico)) reference strain
1 entry
Bean golden yellow mosaic virus (isolate Puerto Rico-Japan) (BGYMV) reference strain
1 entry
Bean yellow dwarf virus (BeYDV) reference strain
1 entry
Beet curly top virus (strain California/Logan) (BCTV) reference strain
1 entry
Borna disease virus (strain V) (BDV) reference strain
1 entry
Bovine adenovirus B serotype 3 (BAdV-3) (Mastadenovirus bos3) reference strain
1 entry
Bovine papillomavirus type 1 reference strain
1 entry
Bovine papillomavirus type 3 reference strain
1 entry
Bovine papillomavirus type 5 reference strain
1 entry
Bovine parvovirus 1 (BPV-1) reference strain
1 entry
Bovine polyomavirus (BPyV) (Bos taurus polyomavirus 1) reference strain
1 entry
Budgerigar fledgling disease virus (BFPyV) (Aves polyomavirus 1) reference strain
1 entry
Cabbage leaf curl virus (isolate Jamaica) (CaLCuV) reference strain
1 entry
Canary circovirus (CaCV) reference strain
1 entry
Canine adenovirus serotype 1 (strain RI261) (CAdV-1) (Canine adenovirus 1 (strain RI261)) reference strain
1 entry
Canine oral papillomavirus (strain Y62) (COPV) reference strain
1 entry
Canine parvovirus type 2 (isolate Dog/United States/CPV-N/1978) (CPV-2) reference strain
1 entry
Carnation etched ring virus (CERV) reference strain
1 entry
Cauliflower mosaic virus (strain Strasbourg) (CaMV) reference strain
1 entry
Cestrum yellow leaf curling virus (CmYLCV) reference strain
1 entry
Chicken anemia virus (isolate Germany Cuxhaven-1) (CAV) reference strain
1 entry
Chloris striate mosaic virus (CSMV) reference strain
1 entry
Commelina yellow mottle virus (CoYMV) reference strain
1 entry
Cottontail rabbit papillomavirus (strain Kansas) (CRPV) (Papillomavirus sylvilagi) reference strain
1 entry
Duck hepatitis B virus (isolate Shanghai/DHBVQCA34) (DHBV) reference strain
1 entry
Epstein-Barr virus (strain AG876) (HHV-4) (Human herpesvirus 4) reference strain
1 entry
Epstein-Barr virus (strain B95-8) (HHV-4) (Human herpesvirus 4) reference strain
1 entry
Equine herpesvirus 1 (strain Ab4p) (EHV-1) (Equine abortion virus) reference strain
1 entry
Equine herpesvirus 2 (strain 86/87) (EHV-2) reference strain
1 entry
European elk papillomavirus (EEPV) reference strain
1 entry
Feline foamy virus (FFV) (Feline syncytial virus) reference strain
1 entry
Figwort mosaic virus (strain DxS) (FMV) reference strain
1 entry
Fowl adenovirus A serotype 1 (strain CELO / Phelps) (FAdV-1) (Avian adenovirus gal1 (strain Phelps)) reference strain
1 entry
Gallid herpesvirus 2 (strain Chicken/Md5/ATCC VR-987) (GaHV-2) (Marek's disease herpesvirus type 1) reference strain
1 entry
Goose circovirus (GoCV) reference strain
1 entry
Ground squirrel hepatitis virus (strain 27) (GSHV) reference strain
1 entry
Hepatitis B virus genotype C subtype ayr (isolate Human/Japan/Okamoto/-) (HBV-C) reference strain
1 entry
Hepatitis B virus genotype D subtype ayw (isolate France/Tiollais/1979) (HBV-D) reference strain
1 entry
Heron hepatitis B virus (HHBV) reference strain
1 entry
Human adenovirus C serotype 2 (HAdV-2) (Human adenovirus 2) reference strain
1 entry
Human adenovirus F serotype 40 (HAdV-40) (Human adenovirus 40) reference strain
1 entry
Human associated cyclovirus 1 (isolate Homo sapiens/Pakistan/PK5510/2007) (HuCyV-1) (Cyclovirus PK5510) reference strain
1 entry
Human cytomegalovirus (strain AD169) (HHV-5) (Human herpesvirus 5) reference strain
1 entry
Human cytomegalovirus (strain Merlin) (HHV-5) (Human herpesvirus 5) reference strain
1 entry
Human herpesvirus 1 (strain 17) (HHV-1) (Human herpes simplex virus 1) reference strain
1 entry
Human herpesvirus 2 (strain HG52) (HHV-2) (Human herpes simplex virus 2) reference strain
1 entry
Human herpesvirus 6A (strain Uganda-1102) (HHV-6 variant A) (Human B lymphotropic virus) reference strain
1 entry
Human herpesvirus 6B (strain Z29) (HHV-6 variant B) (Human B lymphotropic virus) reference strain
1 entry
Human herpesvirus 8 type P (isolate GK18) (HHV-8) (Kaposi's sarcoma-associated herpesvirus) reference strain
1 entry
Human immunodeficiency virus type 1 group M subtype A (isolate U455) (HIV-1) reference strain
1 entry
Human papillomavirus 11 reference strain
1 entry
Human papillomavirus 30 reference strain
1 entry
Human papillomavirus 4 reference strain
1 entry
Human papillomavirus 5 reference strain
1 entry
Human papillomavirus 7 reference strain
1 entry
Human papillomavirus 9 reference strain
1 entry
Human papillomavirus type 1 (Human papillomavirus type 1a) reference strain
1 entry
Human papillomavirus type 10 reference strain
1 entry
Human papillomavirus type 16 reference strain
1 entry
Human papillomavirus type 18 reference strain
1 entry
Human papillomavirus type 26 reference strain
1 entry
Human papillomavirus type 2a reference strain
1 entry
Human papillomavirus type 32 reference strain
1 entry
Human papillomavirus type 34 reference strain
1 entry
Human papillomavirus type 41 reference strain
1 entry
Human papillomavirus type 48 reference strain
1 entry
Human papillomavirus type 49 reference strain
1 entry
Human papillomavirus type 50 reference strain
1 entry
Human papillomavirus type 54 reference strain
1 entry
Human papillomavirus type 60 reference strain
1 entry
Human papillomavirus type 61 reference strain
1 entry
Human papillomavirus type 63 reference strain
1 entry
Human papillomavirus type 6b reference strain
1 entry
Human parvovirus B19 (strain HV) (HPV B19) reference strain
1 entry
Human spumaretrovirus (SFVcpz(hu)) (Human foamy virus) reference strain
1 entry
Indian cassava mosaic virus (ICMV) reference strain
1 entry
Infectious salmon anemia virus (isolate Atlantic salmon/Norway/810/9/99) (ISAV) reference strain
1 entry
Influenza A virus (strain A/Goose/Guangdong/1/1996 H5N1 genotype Gs/Gd) reference strain
1 entry
Influenza A virus (strain A/Puerto Rico/8/1934 H1N1) reference strain
1 entry
Influenza B virus (strain B/Lee/1940) reference strain
1 entry
Influenza C virus (strain C/Ann Arbor/1/1950) reference strain
1 entry
JC polyomavirus (JCPyV) (JCV) reference strain
1 entry
KI polyomavirus (isolate Stockholm 60) (KIPyV) reference strain
1 entry
Macaca mulata papillomavirus 1 (Rhpv 1) (Rhesus papillomavirus type 1) reference strain
1 entry
Maize streak virus genotype A (isolate South Africa) (MSV) reference strain
1 entry
Miscanthus streak virus (isolate 91) (MiSV) reference strain
1 entry
Murine minute virus (strain MVM prototype) (MVM) (Murine minute virus (strain MVM(p))) reference strain
1 entry
Odocoileus virginianus papillomavirus 1 (DPV) (Deer papillomavirus) reference strain
1 entry
Pepper huasteco yellow vein virus (PHYVV) (Pepper huasteco virus) reference strain
1 entry
Pigeon circovirus (PiCV) (Columbid circovirus) reference strain
1 entry
Porcine circovirus 1 (PCV1) reference strain
1 entry
Potato yellow mosaic virus (isolate Venezuela) (PYMV) reference strain
1 entry
Psittacid herpesvirus 1 (isolate Amazon parrot/-/97-0001/1997) (PsHV-1) (Pacheco's disease virus) reference strain
1 entry
Rice tungro bacilliform virus (isolate Philippines) (RTBV) reference strain
1 entry
Saimiriine herpesvirus 2 (strain 11) (SaHV-2) (Herpesvirus saimiri) reference strain
1 entry
Simian foamy virus (isolate chimpanzee) (SFVcpz) reference strain
1 entry
Simian foamy virus type 1 (SFVmac) (SFV-1) reference strain
1 entry
Simian foamy virus type 3 (strain LK3) (SFVagm) (SFV-3) reference strain
1 entry
Simian immunodeficiency virus agm.grivet (isolate AGM gr-1) (SIV-agm.gri) (Simian immunodeficiency virus African green monkey grivet) reference strain
1 entry
Simian virus 40 (SV40) reference strain
1 entry
Snake adenovirus serotype 1 (SnAdV-1) reference strain
1 entry
Soybean chlorotic mottle virus reference strain
1 entry
Squash leaf curl virus (SLCV) reference strain
1 entry
Squirrel monkey polyomavirus reference strain
1 entry
Sugarcane streak virus (isolate South Africa) (SSV) (Sugarcane streak virus (isolate Natal)) reference strain
1 entry
Thogoto virus (isolate SiAr 126) (Tho) reference strain
1 entry
Tobacco yellow dwarf virus (strain Australia) (TYDV) reference strain
1 entry
Tomato golden mosaic virus (strain Yellow vein) (TGMV) reference strain
1 entry
Tomato leaf curl virus (strain Australia) (ToLCV) reference strain
1 entry
Tomato mottle virus (isolate Florida) (ToMoV) reference strain
1 entry
Tomato pseudo-curly top virus (TPCTV) reference strain
1 entry
Tomato yellow leaf curl China virus (TYLCCNV) reference strain
1 entry
Tomato yellow leaf curl Sardinia virus (TYLCSV) reference strain
1 entry
Tomato yellow leaf curl virus (strain Israel) (TYLCV) reference strain
1 entry
Varicella-zoster virus (strain Dumas) (HHV-3) (Human herpesvirus 3) reference strain
1 entry
WU polyomavirus (WUPyV) reference strain
1 entry
Woodchuck hepatitis B virus (isolate 7) (WHV) reference strain
1 entry
Woolly monkey hepatitis B virus (isolate Louisville) (WMHBV) reference strain
2 entries
Hepatitis delta virus genotype I (isolate American) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Italian) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Japanese M-1) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Japanese M-2) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Japanese S-2) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Lebanon-1) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Nauru) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate US-2) (HDV)
2 entries
Hepatitis delta virus genotype I (isolate Woodchuck) (HDV)
2 entries
Hepatitis delta virus genotype II (isolate 7/18/83) (HDV)
2 entries
Hepatitis delta virus genotype II (isolate Japanese S-1) (HDV)
2 entries
Hepatitis delta virus genotype III (isolate Peru-1) (HDV)
2 entries
Hepatitis delta virus genotype III (isolate VnzD8375) (HDV)
2 entries
Hepatitis delta virus genotype III (isolate VnzD8624) (HDV)
2 entries
Human immunodeficiency virus type 1 group M subtype A (isolate MAL) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate ARV2/SF2) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate BH10) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate BH5) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate BRU/LAI) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate JRCSF) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate MN) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate NY5) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate OYI) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate RF/HAT3) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (isolate YU-2) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype B (strain 89.6) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype C (isolate 92BR025) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype C (isolate ETH2220) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype D (isolate ELI) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype D (isolate Z2/CDC-Z34) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype F1 (isolate 93BR020) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype F1 (isolate VI850) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype F2 (isolate MP255) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype F2 (isolate MP257) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype G (isolate 92NG083) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype G (isolate SE6165) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype H (isolate 90CF056) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype H (isolate VI991) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype J (isolate SE9173) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype J (isolate SE9280) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype K (isolate 96CM-MP535) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group M subtype K (isolate 97ZR-EQTB11) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group N (isolate YBF106) (HIV-1)
2 entries
Human immunodeficiency virus type 1 group O (isolate MVP5180) (HIV-1)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate CAM2) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate D194) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate Ghana-1) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate KR) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate NIH-Z) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate ROD) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate SBLISY) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype A (isolate ST) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype B (isolate D205) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype B (isolate EHO) (HIV-2)
2 entries
Human immunodeficiency virus type 2 subtype B (isolate UC1) (HIV-2)
2 entries
Simian immunodeficiency virus (isolate CPZ GAB1) (SIV-cpz) (Chimpanzee immunodeficiency virus)
2 entries
Simian immunodeficiency virus (isolate EK505) (SIV-cpz) (Chimpanzee immunodeficiency virus)
2 entries
Simian immunodeficiency virus (isolate F236/smH4) (SIV-sm) (Simian immunodeficiency virus sooty mangabey monkey)
2 entries
Simian immunodeficiency virus (isolate GB1) (SIV-mnd) (Simian immunodeficiency virus mandrill)
2 entries
Simian immunodeficiency virus (isolate MB66) (SIV-cpz) (Chimpanzee immunodeficiency virus)
2 entries
Simian immunodeficiency virus (isolate Mm142-83) (SIV-mac) (Simian immunodeficiency virus rhesus monkey)
2 entries
Simian immunodeficiency virus (isolate Mm251) (SIV-mac) (Simian immunodeficiency virus rhesus monkey)
2 entries
Simian immunodeficiency virus (isolate PBj14/BCL-3) (SIV-sm) (Simian immunodeficiency virus sooty mangabey monkey)
2 entries
Simian immunodeficiency virus (isolate TAN1) (SIV-cpz) (Chimpanzee immunodeficiency virus)
1 entry
African cassava mosaic virus (isolate Nigerian) (ACMV) (Cassava latent virus (isolate Nigerian))
1 entry
Arctic squirrel hepatitis virus (ASHV)
1 entry
B-lymphotropic polyomavirus (LPV)
1 entry
BK polyomavirus (strain AS) (BKPyV)
1 entry
Beak and feather disease virus (BFDV)
1 entry
Borna disease virus 1 (BoDV-1)
1 entry
Bos taurus papillomavirus 2 (Bovine papillomavirus 2)
1 entry
Bos taurus papillomavirus 4 (Bovine papillomavirus 4)
1 entry
Bovine adenovirus 2 (BAdV-2) (Mastadenovirus bos2)
1 entry
Canine adenovirus serotype 1 (strain CLL) (CAdV-1) (Canine adenovirus 1 (strain CLL))
1 entry
Canine adenovirus serotype 1 (strain Utrecht) (CAdV-1) (Canine adenovirus 1 (strain Utrecht))
1 entry
Canine adenovirus serotype 2 (strain Toronto A 26-61) (CAdV-2) (Canine adenovirus 2 (strain Toronto A 26-61))
1 entry
Canine parvovirus type 2 (CPV-2)
1 entry
Canine parvovirus type 2 (isolate Dog/United States/CPV-b/1978) (CPV-2)
1 entry
Canine parvovirus type 2 (strain Dog/United States/780929/-) (CPV-2)
1 entry
Canine parvovirustype 2 (isolate Dog/United States/CPV-d/1988) (CPV-2)
1 entry
Cauliflower mosaic virus (strain BBC) (CaMV)
1 entry
Cauliflower mosaic virus (strain CM-1841) (CaMV)
1 entry
Cauliflower mosaic virus (strain D/H) (CaMV)
1 entry
Cauliflower mosaic virus (strain NY8153) (CaMV)
1 entry
Chicken anemia virus (isolate Australia) (CAV)
1 entry
Chicken anemia virus (isolate Australia/CAU269-7/2000) (CAV)
1 entry
Chicken anemia virus (isolate Japan 82-2) (CAV)
1 entry
Chicken anemia virus (isolate USA 26p4) (CAV)
1 entry
Chicken anemia virus (isolate USA CIA-1) (CAV)
1 entry
Chimpanzee hepatitis B virus (isolate United Kingdom/LSH/1988) (HBVcpz)
1 entry
Dhori virus (strain Indian/1313/61) (Dho)
1 entry
Duck hepatitis B virus (isolate brown Shanghai duck S5) (DHBV)
1 entry
Duck hepatitis B virus (isolate white Shanghai duck S31) (DHBV)
1 entry
Duck hepatitis B virus (strain China) (DHBV)
1 entry
Duck hepatitis B virus (strain Germany/DHBV-3) (DHBV)
1 entry
Duck hepatitis B virus (strain United States/DHBV-16) (DHBV)
1 entry
Elephantid herpesvirus 1 (isolate Asian elephant/Berlin/Kiba/1998) (EIHV-1) (Elephant endotheliotropic herpesvirus)
1 entry
Epstein-Barr virus (strain GD1) (HHV-4) (Human gammaherpesvirus 4)
1 entry
Equine herpesvirus 1 (strain V592) (EHV-1) (Equine abortion virus)
1 entry
Feline panleukopenia virus (FPV)
1 entry
Feline panleukopenia virus (strain 193) (FPV)
1 entry
Gibbon hepatitis B virus subtype ayw3q (isolate Hope) (HBVgbn)
1 entry
Gorilla hepatitis B virus (isolate Cameroon/gor97) (HBVgor)
1 entry
Hamster parvovirus H1
1 entry
Hamster polyomavirus (HaPyV) (Mesocricetus auratus polyomavirus 1)
1 entry
Hepatitis B virus genotype A1 subtype adw (isolate Philippines/pFDW294/1988) (HBV-A)
1 entry
Hepatitis B virus genotype A1 subtype adw2 (isolate South Africa/84/2001) (HBV-A)
1 entry
Hepatitis B virus genotype A2 (isolate Japan/11D11HCCW/1998) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw (isolate Japan/Nishioka/1983) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw2 (isolate Germany/991/1990) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw2 (strain Rutter 1979) (HBV-A)
1 entry
Hepatitis B virus genotype A3 (isolate Cameroon/CMR711/1994) (HBV-A)
1 entry
Hepatitis B virus genotype A3 (isolate Cameroon/CMR983/1994) (HBV-A)
1 entry
Hepatitis B virus genotype B/C subtype adw (isolate Okinawa/pODW282/1998) (HBV-B)
1 entry
Hepatitis B virus genotype B1 (isolate Japan/Ry30/2002) (HBV-B)
1 entry
Hepatitis B virus genotype B1 (isolate Japan/Yamagata-2/1998) (HBV-B)
1 entry
Hepatitis B virus genotype B1 subtype adw (isolate Japan/pJDW233/1988) (HBV-B)
1 entry
Hepatitis B virus genotype B2 (isolate Indonesia/pIDW420/1988) (HBV-B)
1 entry
Hepatitis B virus genotype B2 (isolate Vietnam/9873/1997) (HBV-B)
1 entry
Hepatitis B virus genotype B2 subtype adw (isolate China/patient4/1996) (HBV-B)
1 entry
Hepatitis B virus genotype C (isolate Vietnam/3270/2000) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ad (isolate Japan/S-179/1988) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Japan/A4/1994) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Japan/Nishioka/1983) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Korea/Kim/1989) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (strain Japan/adr4/1983) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ar (isolate Japan/S-207/1988) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ayw (isolate Australia/AustRC/1992) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ayw (isolate China/Tibet127/2002) (HBV-C)
1 entry
Hepatitis B virus genotype D (isolate France/alpha1/1989) (HBV-D)
1 entry
Hepatitis B virus genotype D (isolate Germany/1-91/1991) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype adw (isolate United Kingdom/adyw/1979) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Australia/AustKW/1991) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Italy/CI/1992) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Japan/JYW796/1988) (HBV-D)
1 entry
Hepatitis B virus genotype F2 (isolate Brazil/w4B) (HBV-F)
1 entry
Hepatitis B virus genotype F2 subtype adw4q (isolate Senegal/9203) (HBV-F)
1 entry
Hepatitis B virus genotype G (isolate IG29227/2000) (HBV-G)
1 entry
Hepatitis B virus genotype G (isolate United States/USG17/2002) (HBV-G)
1 entry
Hepatitis B virus genotype G subtype adw2 (isolate United States/sf/1990) (HBV-G)
1 entry
Hepatitis B virus genotype H subtype adw4 (isolate Nicaragua/2928Nic/1997) (HBV-H)
1 entry
Hepatitis delta virus genotype I (isolate HDV/Human/Central African Republic/FH27/1985) (HDV)
1 entry
Human adenovirus C serotype 5 (HAdV-5) (Human adenovirus 5)
1 entry
Human adenovirus E serotype 4 (HAdV-4) (Human adenovirus 4)
1 entry
Human herpesvirus 7 (strain JI) (HHV-7) (Human T lymphotropic virus)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate HXB3) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate LW123) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate PCV12) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate SC) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate SF33) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate NDK) (HIV-1)
1 entry
Human papillomavirus 12
1 entry
Human papillomavirus 13
1 entry
Human papillomavirus 14
1 entry
Human papillomavirus 15
1 entry
Human papillomavirus 17
1 entry
Human papillomavirus 19
1 entry
Human papillomavirus 20
1 entry
Human papillomavirus 21
1 entry
Human papillomavirus 22
1 entry
Human papillomavirus 23
1 entry
Human papillomavirus 24
1 entry
Human papillomavirus 25
1 entry
Human papillomavirus 27
1 entry
Human papillomavirus 28
1 entry
Human papillomavirus 29
1 entry
Human papillomavirus 3
1 entry
Human papillomavirus 31
1 entry
Human papillomavirus 33
1 entry
Human papillomavirus 35
1 entry
Human papillomavirus 36
1 entry
Human papillomavirus 37
1 entry
Human papillomavirus 38
1 entry
Human papillomavirus 39
1 entry
Human papillomavirus 40
1 entry
Human papillomavirus 42
1 entry
Human papillomavirus 43
1 entry
Human papillomavirus 44
1 entry
Human papillomavirus 45
1 entry
Human papillomavirus 47
1 entry
Human papillomavirus 51
1 entry
Human papillomavirus 52
1 entry
Human papillomavirus 56
1 entry
Human papillomavirus 57
1 entry
Human papillomavirus 58
1 entry
Human papillomavirus 62
1 entry
Human papillomavirus 65
1 entry
Human papillomavirus 66
1 entry
Human papillomavirus 67
1 entry
Human papillomavirus 69
1 entry
Human papillomavirus 82
1 entry
Human papillomavirus type 53
1 entry
Human papillomavirus type 55
1 entry
Human papillomavirus type 5b
1 entry
Human papillomavirus type 6a
1 entry
Human papillomavirus type 70
1 entry
Human papillomavirus type 8
1 entry
Human papillomavirus type ME180
1 entry
Human parvovirus B19 (isolate AU) (HPV B19)
1 entry
Infectious laryngotracheitis virus (strain Thorne V882) (ILTV) (Gallid herpesvirus 1)
1 entry
Influenza A virus (strain A/Anas acuta/Primorje/695/1976 H2N3)
1 entry
Influenza A virus (strain A/Ann Arbor/6/1960 H2N2)
1 entry
Influenza A virus (strain A/Beijing/337/1989 H3N2)
1 entry
Influenza A virus (strain A/Beijing/352/1989 H3N2)
1 entry
Influenza A virus (strain A/Beijing/353/1989 H3N2)
1 entry
Influenza A virus (strain A/Beijing/39/1975 H3N2)
1 entry
Influenza A virus (strain A/Brazil/11/1978 H1N1)
1 entry
Influenza A virus (strain A/Brevig Mission/1/1918 H1N1) (Influenza A virus (strain A/South Carolina/1/1918 H1N1))
1 entry
Influenza A virus (strain A/Budgerigar/Hokkaido/1/1977 H4N6)
1 entry
Influenza A virus (strain A/California/10/1978 H1N1)
1 entry
Influenza A virus (strain A/Chicken/Germany/n/1949 H10N7)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/31.2/2002 H5N1 genotype X1)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/37.4/2002 H5N1 genotype X2)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/715.5/2001 H5N1 genotype E)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/96.1/2002 H5N1 genotype Y)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/FY150/2001 H5N1 genotype D)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/YU22/2002 H5N1 genotype Z)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/YU562/2001 H5N1 genotype B)
1 entry
Influenza A virus (strain A/Chicken/Pennsylvania/1/1983 H5N2)
1 entry
Influenza A virus (strain A/Chicken/Scotland/1959 H5N1)
1 entry
Influenza A virus (strain A/Chicken/Shantou/4231/2003 H5N1 genotype V)
1 entry
Influenza A virus (strain A/Chile/1/1983 H1N1)
1 entry
Influenza A virus (strain A/China:Nanchang/11/1996 H1N1)
1 entry
Influenza A virus (strain A/Duck/Australia/749/1980 H1N1)
1 entry
Influenza A virus (strain A/Duck/Bavaria/2/1977 H1N1)
1 entry
Influenza A virus (strain A/Duck/Beijing/1/1978 H3N6)
1 entry
Influenza A virus (strain A/Duck/Czechoslovakia/1956 H4N6)
1 entry
Influenza A virus (strain A/Duck/England/1/1956 H11N6)
1 entry
Influenza A virus (strain A/Duck/England/1/1962 H4N6)
1 entry
Influenza A virus (strain A/Duck/Germany/1949 H10N7)
1 entry
Influenza A virus (strain A/Duck/Hokkaido/8/1980 H3N8)
1 entry
Influenza A virus (strain A/Duck/Hong Kong/2986.1/2000 H5N1 genotype C)
1 entry
Influenza A virus (strain A/Duck/Hong Kong/7/1975 H3N2)
1 entry
Influenza A virus (strain A/Duck/Manitoba/1/1953 H10N7)
1 entry
Influenza A virus (strain A/Duck/Memphis/928/1974 H3N8)
1 entry
Influenza A virus (strain A/Duck/New Zealand/31/1976 H4N6)
1 entry
Influenza A virus (strain A/Duck/Pennsylvania/1/1969 H6N1)
1 entry
Influenza A virus (strain A/Duck/Ukraine/2/1960 H11N8)
1 entry
Influenza A virus (strain A/England/19/1955 H1N1)
1 entry
Influenza A virus (strain A/Equine/Jillin/1/1989 H3N8)
1 entry
Influenza A virus (strain A/Equine/Kentucky/2/1986 H3N8)
1 entry
Influenza A virus (strain A/Equine/London/1416/1973 H7N7)
1 entry
Influenza A virus (strain A/Equine/Miami/1/1963 H3N8)
1 entry
Influenza A virus (strain A/Equine/Prague/1/1956 H7N7)
1 entry
Influenza A virus (strain A/Equine/Santiago/1/1985 H3N8)
1 entry
Influenza A virus (strain A/Equine/Tennessee/5/1986 H3N8)
1 entry
Influenza A virus (strain A/Fort Monmouth/1/1947 H1N1)
1 entry
Influenza A virus (strain A/Fort Warren/1/1950 H1N1)
1 entry
Influenza A virus (strain A/Fowl plague virus/Dobson/'Dutch'/1927 H7N7)
1 entry
Influenza A virus (strain A/Fowl plague virus/Rostock/8/1934 H7N1)
1 entry
Influenza A virus (strain A/Grey teal/Australia/2/1979 H4N4)
1 entry
Influenza A virus (strain A/Guangdong/38/1977 H3N2)
1 entry
Influenza A virus (strain A/Guangdong/39/1989 H3N2)
1 entry
Influenza A virus (strain A/Guangdong/9/1987 H3N2)
1 entry
Influenza A virus (strain A/Guinea fowl/Hong Kong/38/2002 H5N1 genotype X0)
1 entry
Influenza A virus (strain A/Gull/Astrakhan/227/1984 H13N6)
1 entry
Influenza A virus (strain A/Gull/Maryland/1815/1979 H13N6)
1 entry
Influenza A virus (strain A/Gull/Maryland/1824/1978 H13N6)
1 entry
Influenza A virus (strain A/Gull/Maryland/5/1977 H11N9)
1 entry
Influenza A virus (strain A/Gull/Maryland/704/1977 H13N6)
1 entry
Influenza A virus (strain A/Gull/Massachusetts/26/1980 H13N6)
1 entry
Influenza A virus (strain A/Gull/Minnesota/945/1980 H13N6)
1 entry
Influenza A virus (strain A/Henry/1936 H1N1)
1 entry
Influenza A virus (strain A/Hickox/1940 H1N1)
1 entry
Influenza A virus (strain A/Hong Kong/1/1968 H3N2)
1 entry
Influenza A virus (strain A/Hong Kong/156/1997 H5N1 genotype Gs/Gd)
1 entry
Influenza A virus (strain A/Hong Kong/212/2003 H5N1 genotype Z+)
1 entry
Influenza A virus (strain A/Hong Kong/5/1983 H3N2)
1 entry
Influenza A virus (strain A/India/6263/1980 H1N1)
1 entry
Influenza A virus (strain A/Kiev/59/1979 H1N1)
1 entry
Influenza A virus (strain A/Kitakyushu/159/1993 H3N2)
1 entry
Influenza A virus (strain A/Leningrad/1/1954 H1N1)
1 entry
Influenza A virus (strain A/Malaysia:Malaya/302/1954 H1N1)
1 entry
Influenza A virus (strain A/Mallard/Astrakhan/244/1982 H14N6) (Influenza A virus (strain A/Mallard/Gurjev/244/1982 H14N6))
1 entry
Influenza A virus (strain A/Mallard/Astrakhan/263/1982 H14N5) (Influenza A virus (strain A/Mallard/Gurjev/263/1982 H14N5))
1 entry
Influenza A virus (strain A/Mallard/New York/6750/1978 H2N2)
1 entry
Influenza A virus (strain A/Memphis/1/1971 H3N2)
1 entry
Influenza A virus (strain A/Memphis/1/1973 H3N2)
1 entry
Influenza A virus (strain A/Memphis/101/1972 H3N2)
1 entry
Influenza A virus (strain A/Memphis/110/1976 H3N2)
1 entry
Influenza A virus (strain A/Memphis/14/1985 H3N2)
1 entry
Influenza A virus (strain A/Memphis/18/1978 H3N2)
1 entry
Influenza A virus (strain A/Memphis/2/1978 H3N2)
1 entry
Influenza A virus (strain A/Memphis/3/1988 H3N2)
1 entry
Influenza A virus (strain A/Memphis/4/1973 H3N2)
1 entry
Influenza A virus (strain A/Memphis/4/1980 H3N2)
1 entry
Influenza A virus (strain A/Memphis/5/1980 H3N2)
1 entry
Influenza A virus (strain A/Memphis/6/1986 H3N2)
1 entry
Influenza A virus (strain A/Memphis/8/1988 H3N2)
1 entry
Influenza A virus (strain A/Mink/Sweden/1984 H10N4)
1 entry
Influenza A virus (strain A/New Jersey/8/1976 H1N1)
1 entry
Influenza A virus (strain A/New Zealand:South Canterbury/35/2000 H1N1)
1 entry
Influenza A virus (strain A/Northern Territory/60/1968 H3N2) (Influenza A virus (strain NT60)) (Influenza A virus (strain A/NT/60/1968 H3N2))
1 entry
Influenza A virus (strain A/Ohio/4/1983 H1N1)
1 entry
Influenza A virus (strain A/Parrot/Ulster/1973 H7N1)
1 entry
Influenza A virus (strain A/Port Chalmers/1/1973 H3N2)
1 entry
Influenza A virus (strain A/Ruddy Turnstone/New Jersey/47/1985 H4N6)
1 entry
Influenza A virus (strain A/Russia:St.Petersburg/8/2006 H1N1)
1 entry
Influenza A virus (strain A/Seal/Massachusetts/1/1980 H7N7)
1 entry
Influenza A virus (strain A/Shanghai/11/1987 H3N2)
1 entry
Influenza A virus (strain A/Shanghai/16/1989 H3N2)
1 entry
Influenza A virus (strain A/Shanghai/31/1980 H3N2)
1 entry
Influenza A virus (strain A/Shanghai/6/1990 H3N2)
1 entry
Influenza A virus (strain A/Shearwater/Australia/1972 H6N5)
1 entry
Influenza A virus (strain A/Silky Chicken/Hong Kong/SF189/2001 H5N1 genotype A)
1 entry
Influenza A virus (strain A/Silky Chicken/Hong Kong/YU100/2002 H5N1 genotype X3)
1 entry
Influenza A virus (strain A/Singapore/1/1957 H2N2)
1 entry
Influenza A virus (strain A/Swine/29/1937 H1N1)
1 entry
Influenza A virus (strain A/Swine/41/1949 H1N1)
1 entry
Influenza A virus (strain A/Swine/Cambridge/1/1935 H1N1)
1 entry
Influenza A virus (strain A/Swine/Colorado/1/1977 H3N2)
1 entry
Influenza A virus (strain A/Swine/Dandong/9/1983 H3N2)
1 entry
Influenza A virus (strain A/Swine/Germany/2/1981 H1N1)
1 entry
Influenza A virus (strain A/Swine/Hong Kong/126/1982 H3N2)
1 entry
Influenza A virus (strain A/Swine/Hong Kong/127/1982 H3N2)
1 entry
Influenza A virus (strain A/Swine/Hong Kong/6/1976 H3N2)
1 entry
Influenza A virus (strain A/Swine/Iowa/15/1930 H1N1)
1 entry
Influenza A virus (strain A/Swine/Iowa/17672/1988 H1N1)
1 entry
Influenza A virus (strain A/Swine/Iowa/1946 H1N1)
1 entry
Influenza A virus (strain A/Swine/Iowa/1976/1931 H1N1)
1 entry
Influenza A virus (strain A/Swine/Italy/141/1981 H1N1)
1 entry
Influenza A virus (strain A/Swine/Italy/2/1979 H1N1)
1 entry
Influenza A virus (strain A/Swine/Italy/437/1976 H1N1)
1 entry
Influenza A virus (strain A/Swine/Italy/839/1989 H1N1)
1 entry
Influenza A virus (strain A/Swine/Jamesburg/1942 H1N1)
1 entry
Influenza A virus (strain A/Swine/May/1954 H1N1)
1 entry
Influenza A virus (strain A/Swine/Netherlands/12/1985 H1N1)
1 entry
Influenza A virus (strain A/Swine/Ohio/23/1935 H1N1)
1 entry
Influenza A virus (strain A/Swine/Ontario/2/1981 H1N1)
1 entry
Influenza A virus (strain A/Swine/Tennessee/24/1977 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1957 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1961 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1967 H1N1)
1 entry
Influenza A virus (strain A/Teal/China/2978.1/2002 H5N1 genotype W)
1 entry
Influenza A virus (strain A/Teal/Iceland/29/1980 H7N7)
1 entry
Influenza A virus (strain A/Tern/South Africa/1961 H5N3)
1 entry
Influenza A virus (strain A/Tern/Turkmenia/18/1972 H3N3)
1 entry
Influenza A virus (strain A/Texas/1/1977 H3N2)
1 entry
Influenza A virus (strain A/Turkey/Ireland/1378/1983 H5N8)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/1661/1981 H1N1)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/833/1980 H4N2)
1 entry
Influenza A virus (strain A/Turkey/Ontario/6118/1968 H8N4)
1 entry
Influenza A virus (strain A/Turkey/Ontario/7732/1966 H5N9)
1 entry
Influenza A virus (strain A/Turkey/Wisconsin/1/1966 H9N2)
1 entry
Influenza A virus (strain A/USA:Albany/12/1951 H1N1)
1 entry
Influenza A virus (strain A/USA:Huston/AA/1945 H1N1)
1 entry
Influenza A virus (strain A/USA:Iowa/1943 H1N1)
1 entry
Influenza A virus (strain A/USA:Memphis/10/1996 H1N1)
1 entry
Influenza A virus (strain A/USA:Phila/1935 H1N1)
1 entry
Influenza A virus (strain A/USA:Texas/UR06-0195/2007 H1N1)
1 entry
Influenza A virus (strain A/USSR/90/1977 H1N1)
1 entry
Influenza A virus (strain A/Udorn/307/1972 H3N2)
1 entry
Influenza A virus (strain A/Victoria/5/1968 H2N2)
1 entry
Influenza A virus (strain A/Whale/Maine/328/1984 H13N2)
1 entry
Influenza A virus (strain A/Whale/Pacific ocean/19/1976 H1N3)
1 entry
Influenza A virus (strain A/Wilson-Smith/1933 H1N1) (Influenza A virus (strain A/WS/1933 H1N1))
1 entry
Influenza A virus (strain A/Wisconsin/3523/1988 H1N1)
1 entry
Influenza B virus (strain B/Ann Arbor/1/1966 [cold-adapted])
1 entry
Influenza B virus (strain B/Ann Arbor/1/1966 [wild-type])
1 entry
Influenza B virus (strain B/Ann Arbor/1/1986)
1 entry
Influenza B virus (strain B/Panama/45/1990)
1 entry
Influenza B virus (strain B/Singapore/222/1979)
1 entry
Influenza C virus (strain C/California/1978)
1 entry
Influenza C virus (strain C/Johannesburg/1/1966)
1 entry
KI polyomavirus (isolate Stockholm 350) (KIPyV)
1 entry
KI polyomavirus (isolate Stockholm 380) (KIPyV)
1 entry
Maize streak virus genotype A (isolate Kenya) (MSV)
1 entry
Maize streak virus genotype A (isolate Nigeria) (MSV)
1 entry
Maize streak virus genotype B (isolate Tas) (MSV)
1 entry
Maize streak virus genotype C (isolate Set) (MSV)
1 entry
Maize streak virus genotype D (isolate Raw) (MSV)
1 entry
Maize streak virus genotype E (isolate Pat) (MSV)
1 entry
Mungbean yellow mosaic virus (strain Vigna) (MYMV)
1 entry
Murine adenovirus A serotype 1 (MAdV-1) (Murine adenovirus 1)
1 entry
Murine minute virus (strain MVMi) (MVM) (Murine parvovirus)
1 entry
Murine polyomavirus (strain A2) (MPyV)
1 entry
Murine polyomavirus (strain A3) (MPyV)
1 entry
Murine polyomavirus (strain Crawford small-plaque) (MPyV)
1 entry
Murine polyomavirus (strain Kilham) (MPyV) (Murine pneumotropic virus)
1 entry
Panicum streak virus (isolate Kenya) (PanSV)
1 entry
Parvovirus LuIII
1 entry
Porcine circovirus 2 (PCV2)
1 entry
Porcine parvovirus (strain 90HS) (PPV)
1 entry
Porcine parvovirus (strain Kresse) (PPV)
1 entry
Porcine parvovirus (strain NADL-2) (PPV)
1 entry
Pygmy chimpanzee papillomavirus type 1 (PCPV-1)
1 entry
Simian immunodeficiency virus (isolate K78) (SIV-mac) (Simian immunodeficiency virus rhesus monkey)
1 entry
Simian immunodeficiency virus agm.vervet (isolate AGM TYO-1) (SIV-agm.ver) (Simian immunodeficiency virus African green monkey vervet)
1 entry
Simian immunodeficiency virus agm.vervet (isolate AGM155) (SIV-agm.ver) (Simian immunodeficiency virus African green monkey vervet)
1 entry
Simian immunodeficiency virus agm.vervet (isolate AGM3) (SIV-agm.ver) (Simian immunodeficiency virus African green monkey vervet)
1 entry
Simian virus 12 (strain wt100) (SV-12) (Baboon polyomavirus 1)
1 entry
Tomato yellow leaf curl Sardinia virus (isolate Spain-1) (TYLCSV)
1 entry
Tomato yellow leaf curl Sardinia virus (isolate Spain-2) (TYLCSV)
1 entry
Wheat dwarf virus (isolate Sweden) (WDV)
1 entry
Woodchuck hepatitis B virus (isolate 1) (WHV)
1 entry
Woodchuck hepatitis B virus (isolate 2) (WHV)
1 entry
Woodchuck hepatitis B virus (isolate 59) (WHV)
1 entry
