Simplexvirus (taxid:10294)
VIRION
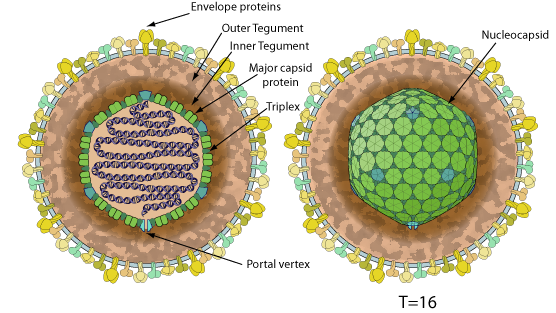
Enveloped, spherical to pleomorphic, 150-200 nm in diameter, T=16 icosahedral symmetry. The capsid consists of 162 capsomers and is surrounded by an amorphous tegument.
GENOME
Monopartite, linear, dsDNA genome of 152 kb. The genome contains terminal and internal reiterated sequences.
GENE EXPRESSION
All genes are transcribed by the host RNA polymerase II. and most mRNAs are unspliced. There are three temporal classes of genes: immediate-early (alpha), early (beta) and late (gamma). The immediate-early genes are transcribed immediately after infection to take control of cell defense and to activate early genes. These encode the proteins necessary for the viral DNA replication. The late genes mostly encode structural proteins. Latent genes can stop the replicative process at the early step. Certain proteins are downregulated translationaly by a leaky scanning from an upstream ORF.
ENZYMES
- DNA-dependent DNA polymerase [UL30]
- DNA primase [UL52]
- Tegument deneddylase (Peptidase C76) [UL36]
- Assemblin (Peptidase S21) [UL26]
- Kinase [UL13]
- Helicase [UL5]
- Endonuclease [UL41]
- Ribonucleoside-diphosphate reductase [UL40]
- Thymidine kinase [UL23]
- Uracil-DNA glycosylase [UL2]
REPLICATION
NUCLEAR
Lytic replication:
- Attachment of the viral gB, gC, gD and gH proteins to host receptors mediates endocytosis of the virus into the host cell.
- Fusion with the plasma membrane to release the core and the tegument proteins into the host cytoplasm.
- The capsid is transported to the nuclear pore where viral DNA is released into the nucleus.
- Transcription of immediate early genes which promote transcription of early genes and protect the virus against innate host immunity.
- Transcription of early viral mRNA by host polymerase II, encoding proteins involved in replication of the viral DNA.
- A first round of circular genome amplification occurs by bidirectional replication
- Synthesis of linear concatemer copies of viral DNA by rolling circle.
- Transcription of late mRNAs by host polymerase II, encoding structural proteins.
- Assembly of the virus in nuclear viral factories and budding through the inner lamella of the nuclear membrane which has been modified by the insertion of herpes glycoproteins, throughout the Golgi and final release at the plasma membrane.
Latent replication : replication of circular viral episome in tandem with the host cell DNA using the host cell replication machinery.
Host-virus interaction
Adaptive immune response inhibition
Herpes simplex protein US12 binds specifically to transporters associated with antigen processing (TAP), blocking peptide-binding to TAP and subsequent loading of peptides onto MHC class I molecules  .
Herpes simplex virus 1 glycoprotein B and US3 collaborate to inhibit CD1d antigen presentation and NKT cell function
.
Herpes simplex virus 1 glycoprotein B and US3 collaborate to inhibit CD1d antigen presentation and NKT cell function  .
.
Apoptosis modulation
The viral kinase US3 modulates host apoptosis in different ways. It activates host protein kinase A/PKA to block apoptosis. Additionally, it also interacts with programmed cell death protein 4 (PDCD4) and retains it in the nucleus thereby inhibiting host apoptosis  .
.
Autophagy modulation
Herpes simplex virus ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein and thus blocking host autophagy process  .
.
Cell-cycle modulation
The viral proteins ICP27 and vhs act jointly to repress host gene transcription and blocks the cell cycle at the G1 phase  . In addition the UL24 protein induces a cell cycle arrest at G2/M transition through inactivation of the host cyclinB/cdc2 complex
. In addition the UL24 protein induces a cell cycle arrest at G2/M transition through inactivation of the host cyclinB/cdc2 complex  .
.
Innate immune response inhibition
Herpes simplex virus inhibits the cascade leading to production of interferon-beta by targeting different cellular proteins. ICP34.5 forms a complex with host TBK1 and thus disrupts the interaction of TBK1-containing complexes with IRF3
 . Another viral protein, ICP0, prevents IRF3 activation and subsequent induction of IFN-beta expression
. Another viral protein, ICP0, prevents IRF3 activation and subsequent induction of IFN-beta expression
 .
.
Host splicing inhibition
HSV-1 ICP27 is an alternative splicing regulator of host mRNA. It has been shown to act as a splicing silencer at the 3' splice site of the PML intron 7a  .
.
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)230 entries grouped by strain
73 entries
Human herpesvirus 1 (strain 17) (HHV-1) (Human herpes simplex virus 1) reference strain
72 entries
Human herpesvirus 2 (strain HG52) (HHV-2) (Human herpes simplex virus 2) reference strain
2 entries
Cercopithecine herpesvirus 1 (strain E2490) (CeHV-1) (Simian herpes B virus) reference strain
15 entries
Human herpesvirus 1 (strain F) (HHV-1) (Human herpes simplex virus 1)
12 entries
Human herpesvirus 1 (strain KOS) (HHV-1) (Human herpes simplex virus 1)
12 entries
Human herpesvirus 2 (strain 333) (HHV-2) (Human herpes simplex virus 2)
6 entries
Human herpesvirus 1 (strain HFEM) (HHV-1) (Human herpes simplex virus 1)
3 entries
Bovine herpesvirus 2 (strain BMV) (BoHV-2) (Bovine mammillitis virus)
3 entries
Cercopithecine herpesvirus 1 (CeHV-1) (Simian herpes B virus)
3 entries
Human herpesvirus 1 (strain Angelotti) (HHV-1) (Human herpes simplex virus 1)
3 entries
Human herpesvirus 1 (strain MP) (HHV-1) (Human herpes simplex virus 1)
3 entries
Human herpesvirus 2 (strain G) (HHV-2) (Human herpes simplex virus 2)
2 entries
Herpes simplex virus type 2 (strain SA8) (Simian agent 8)
2 entries
Human herpesvirus 1 (strain Patton) (HHV-1) (Human herpes simplex virus 1)
2 entries
Human herpesvirus 1 (strain SC16) (HHV-1) (Human herpes simplex virus 1)
2 entries
Human herpesvirus 2 (HHV-2) (Human herpes simplex virus 2)
2 entries
Saimiriine herpesvirus 1 (strain MV-5-4-PSL) (SaHV-1) (Marmoset herpesvirus)
1 entry
Bovine herpesvirus 2 (strain BHM-1) (BoHV-2) (Bovine mammillitis virus)
1 entry
Cercopithecine herpesvirus 16 (CeHV-16) (Herpesvirus papio 2)
1 entry
Herpesvirus ateles type 1 (strain Lennette)
1 entry
Human herpesvirus 1 (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain A44) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain CL101) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain CVG-2) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain HZT) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain MGH-10) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain R15) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 1 (strain R19) (HHV-1) (Human herpes simplex virus 1)
1 entry
Human herpesvirus 2 (strain 186) (HHV-2) (Human herpes simplex virus 2)
1 entry
Human herpesvirus 2 (strain B4327UR) (HHV-2) (Human herpes simplex virus 2)
Cercopithecine alphaherpesvirus 2 taxid:10317
Cercopithecine herpesvirus 1 taxid:10325
Cercopithecine herpesvirus 1 (strain E2490) taxid:260965
| Protein | ModelArchive |
| ICP47 protein (Immediate-early protein IE12) (Immediate-early-5) (Infected cell protein 47) (US12 protein)... | ma-jd-viral-62159 |
Cercopithecine herpesvirus 16 taxid:340907
Chimpanzee herpesvirus strain 105640 taxid:332937
Human herpesvirus 1 taxid:10298
Human herpesvirus 1 (strain 17) taxid:10299
Human herpesvirus 1 (strain MP) taxid:10307
| Protein | ModelArchive |
| Envelope glycoprotein K (Syncytial protein) | ma-jd-viral-49381 |
Human herpesvirus 1 (strain R15) taxid:36345
| Protein | ModelArchive |
| Envelope glycoprotein K (Syncytial protein) | ma-jd-viral-49381 |
Human herpesvirus 2 taxid:10310
Human herpesvirus 2 (strain 333) taxid:10313
Human herpesvirus 2 (strain G) taxid:10314
| Protein | ModelArchive |
| Virion host shutoff protein (Vhs) (EC 3.1.27.-) | ma-jd-viral-22425 |
