TEGUMENT FORMATION
Several studies have characterized the tegument content of different herpesviruses including herpes simplex 1, human cytomegalovirus  , Epstein-Barr virus, or Kaposi virus. The outcome of theses works is the characterization of commun and individual components.
, Epstein-Barr virus, or Kaposi virus. The outcome of theses works is the characterization of commun and individual components.
Inner tegument
The initial steps of the tegument formation seem shared by all herpesviruses.
Three majors events occur to ensure proper tegument folding and correct glycoprotein incorporation into virions.
1- Incorporation of the major tegument proteins UL36 and UL37
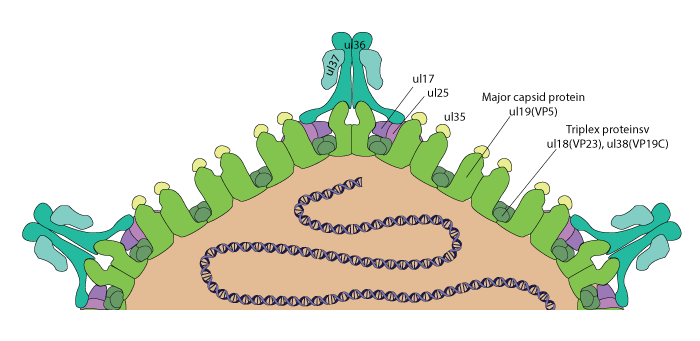
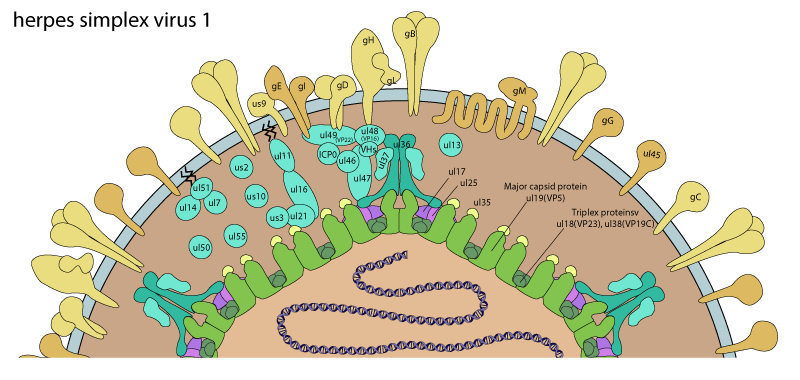
The inner tegument contains the protein UL36 with is associated to the capsid at the vertices by binding to the the UL19-UL25 complex which is termed the C-capsid specific component (CCSC) . This CCSC complex interacts only with the triplexes located at the pentons (vertices)/hexons interfaces. Moreover, this interaction occurs only when the DNA is encapsidated and not before. For this reason, the incorporation of the UL36 tegument protein occurs after DNA packaging. Subsequently, the tegument protein UL37 directly binds to UL36 and is added to the tegument.
. This CCSC complex interacts only with the triplexes located at the pentons (vertices)/hexons interfaces. Moreover, this interaction occurs only when the DNA is encapsidated and not before. For this reason, the incorporation of the UL36 tegument protein occurs after DNA packaging. Subsequently, the tegument protein UL37 directly binds to UL36 and is added to the tegument.
2- The essential UL11-UL16 interaction
The HHV-1 UL16 is another tegument protein that binds to the capsid. Essential for assembly and budding processes, it recognizes UL11 through an acidic-cluster motif, which is located in the first half of the protein. This interaction is essential for viral replication and occurs in different herpeviruses such as HHV-1, varicella virus or HCMV


 . UL11 is associated to cytoplasmic membranes and could therefore brings the capsid to these membranes through UL16 in order to achieve secondary envelopment.
. UL11 is associated to cytoplasmic membranes and could therefore brings the capsid to these membranes through UL16 in order to achieve secondary envelopment.
3- The UL7-UL51 interaction
The UL7 and UL51 proteins are shared by all herpesviruses, and the interaction between these two tegument proteins plays also an important role in the cytoplasmic envelopment of the virions. As membrane associated, UL51 is required for recruitment of UL7 to cytoplasmic membranes in infected cells
 .
.
Outer tegument
In addition to these proteins, the tegument contains additional components that can be either shared by all herpesviruses or specific to each virus.
1- Different proteins for the same purpose
Early modulation of PML bodies and the inhibition of various of its components seems to be a required step for all herpesviruses. As shown in the table, herpes simplex E3 ligase ICP0 inhibits PML bodies and send PML to degradation together with host SP100. HCMV possesses at least two tegument proteins to interfere with PML bodies, UL35 and UL82. Epstein-Barr virus BNRF1 and HHV-8 ORF75 acts on PML bodies and inhibit host DAXX and ATRX, SP100 respectively.
2- Specific tegument components
The tegument of each herpes virus contains specific components that serves specifically their purpose, according to the infected cell type or the viral special needs. For example, herpes simplex virus 1 contains in its tegument a module composed of a virion host shutoff and several of its regulators.
Susan M. Varnum, Daniel N. Streblow, Matthew E. Monroe, Patricia Smith, Kenneth J. Auberry, Ljiljana Pasa-Tolic, Dai Wang, David G. Camp, Karin Rodland, Steven Wiley, William Britt, Thomas Shenk, Richard D. Smith, Jay A. Nelson
J. Virol. October 2004; 78: 10960-10966
David G. Meckes, Jacob A. Marsh, John W. Wills
Virology March 15, 2010; 398: 208?213
Tomohiko Sadaoka, Satoshi Serada, Junko Kato, Mayuko Hayashi, Yasuyuki Gomi, Tetsuji Naka, Koichi Yamanishi, Yasuko Mori
J. Virol. January 2014; 88: 188?201
Stacia L. Phillips, Daniel Cygnar, Alexandra Thomas, Wade A. Bresnahan
J. Virol. September 2012; 86: 9995?10005
Wan H. Fan, Ashley P. E. Roberts, Marion McElwee, David Bhella, Frazer J. Rixon, Rebecca Lauder
J. Virol. February 2015; 89: 1502?1511
Richard J. Roller, Rachel Fetters
J. Virol. March 2015; 89: 3112?3122
