Mastadenovirus (taxid:10509)
VIRION
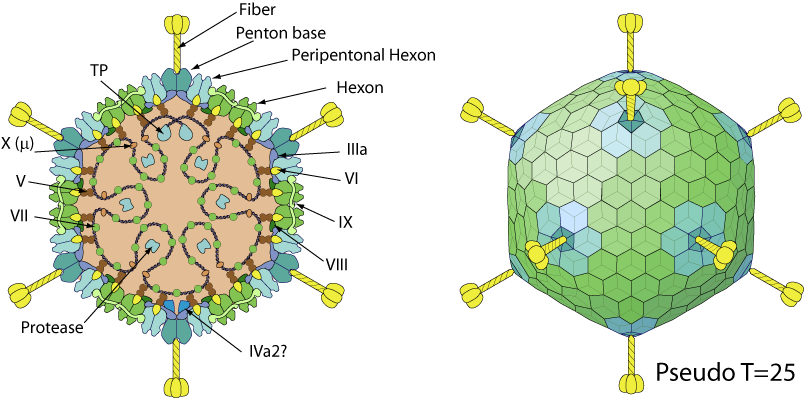
Non-enveloped capsid with a pseudo T=25 icosahedral symmetry 
 . The capsid diameter is about 90 nm. The capsid shell consists of 720 hexon subunits arranged as 240 trimers and 12 vertex penton capsomers each with a fiber protruding from the surface.
. The capsid diameter is about 90 nm. The capsid shell consists of 720 hexon subunits arranged as 240 trimers and 12 vertex penton capsomers each with a fiber protruding from the surface.
- Double jelly roll-fold major capsid protein [Hexon]
- Single jelly roll-fold penton capsid protein [Penton]
GENOME
Monopartite, linear double-stranded DNA of 35-36kb encoding about 40 proteins. The genome has terminally redundant sequences which have inverted terminal repetitions (ITR). The terminal protein (TP) is covalently attached to each end of the genome.
GENE EXPRESSION
Transcription is nuclear, in three phases; early, intermediate (replication), and late (virion assembly). All genes are transcribed by host RNA pol II except virus-associated (VA) gene(s) of some primate adenoviruses transcribed by RNA pol III. Genes transcribed by RNA pol II give rise to multiple mRNA that are produced by alternative splicing and use of different poly(A) sites.
ENZYMES
- DNA-directed DNA polymerase B [POL]
- Protein-primed terminal transferase [POL on TP]
- Adenain (Peptidase C5) [Protease]
REPLICATION
NUCLEUS
- Attachment of the viral fibers to the host CAR adhesion receptor. Subsequent binding of the penton protein to host integrin entry receptors mediates internalization into the host cell by clathrin-mediated endocytosis of the virus and fiber shedding. Some serotypes also seem to use macropinocytosis.
- Disruption of host endosomal membrane by lytic protein VI releases the viral capsid in the cytosol.
- Microtubular transport toward nucleus of the viral genome still protected by the core protein VII and a partial capsid mainly composed of hexons and protein IX.
- Docking at the NPC and capsid disruption.
- Import of the viral genome into host nucleus mediated by core protein VII.
- Transcription of early genes (E genes) by host RNA pol II: these proteins optimize the cellular milieu for viral replication, and counteract a variety of antiviral defenses.
- Intermediate genes activate replication of the DNA genome by DNA strand displacement in the nucleus.
- Expression of L4-22K and L4-33K causes early to late switch. Transcription of late genes (L genes) by host RNA pol II, mostly encoding structural proteins.
- Host translation shutoff performed by the viral 100K protein.
- Assembly of new virions in the nucleus.
- Virions are released by lysis of the cell.
- Virion maturation by the viral protease.
Host-virus interaction
Adaptive immune response inhibition
The adenovirus E19 protein downregulates cell-surface expression of MHC-I by binding and retains the heavy chain in ER. E19 binds TAP and acts as a tapasin inhibitor, preventing class I/TAP association
 .
.
Apoptosis modulation
Soon after infection, the expression of E1A protein drives the cell cycle into S-phase so that the virus can replicate its genome optimally  . However, this arrest leads to the accumulation of host p53 protein and subsequently apoptosis. The apoptosis is then inhibited by different viral proteins including E1B 55K, E4 orf6 or the bcl-2 homolog E1B 19K
. However, this arrest leads to the accumulation of host p53 protein and subsequently apoptosis. The apoptosis is then inhibited by different viral proteins including E1B 55K, E4 orf6 or the bcl-2 homolog E1B 19K  .
.
Autophagy modulation
Autophagy is triggered after adenoviral infection and may facilitate viral replication and/or cell lysis. The E1B 19K viral protein interacts with host Beclin 1, removes Bcl-2 (a negative regulator of autophagy) from Beclin-1 and thereby promotes autophagy  .
.
Cell-cycle modulation
The expression of E1A protein drives the cell cycle into S-phase so that the virus can replicate its genome optimally  .
.
Innate immune response inhibition
Adenoviruses do not inhibit the cascade leading to production of interferon-beta directly. However, they evade the host innate immunity by targeting a cellular histone modification. Indeed, the E1A viral protein blocks the IFN-induced H2B mono-ubiquitination and thereby associated ISGs expression
 .
.
Hongrong Liu, Lei Jin, Sok Boon S. Koh, Ivo Atanasov, Stan Schein, Lily Wu, Z. Hong Zhou
Science August 27, 2010; 329: 1038-1043
Carmen San Mart?n
Viruses May 2012; 4: 847-877
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)336 entries grouped by protein
1 entry
Adenovirus death protein (ADP) (Early-3 11.6 kDa glycoprotein) (E3-11.6K)
7 entries
Pre-hexon-linking protein IIIa (Capsid vertex-specific component IIIa) (CVSC) (Protein IIIa) (pIIIa)
9 entries
Pre-protein VI (pVI)
14 entries
Pre-hexon-linking protein VIII (Pre-protein VIII) (pVIII)
12 entries
Hexon-interlacing protein IX (Protein IX)
14 entries
Hexon protein (CP-H) (Protein II)
7 entries
Penton protein (CP-P) (Penton base protein) (Protein III)
2 entries
Pre-core protein X (pX) (11 kDa core protein) (Protein mu) (pMu)
9 entries
Core-capsid bridging protein (Core protein V)
9 entries
DNA-binding protein (DBP) (Early 2A protein) (Early E2A DNA-binding protein)
2 entries
Probable DNA-binding protein (Agnoprotein)
10 entries
DNA polymerase (EC 2.7.7.7)
1 entry
Early E1A 11 kDa protein
1 entry
Early E1A 18 kDa protein
14 entries
Early E1A protein (Early E1A 20 kDa protein)
11 entries
E1B 55 kDa protein (E1B-55K) (E1B protein, large T-antigen) (E1B-495R)
1 entry
Early E1B 9 kDa protein
15 entries
E1B protein, small T-antigen (E1B 19 kDa protein) (E1B-19K)
1 entry
Early E3 6.4 kDa protein
5 entries
Early E3B 10.4 kDa protein
4 entries
Early E3 7.7 kDa protein
4 entries
Early E3A 12.1 kDa protein
4 entries
Early E3 13.3 kDa protein
6 entries
Early E3 14.5 kDa protein
1 entry
Early E3 16 kDa glycoprotein
4 entries
Early E3 20.3 kDa glycoprotein
5 entries
Early E3 20.5 kDa glycoprotein
2 entries
Early E3 22.1 kDa glycoprotein
1 entry
Early E3 25 kDa glycoprotein
5 entries
Early E3B 12.7 kDa protein
1 entry
Early 3 Conserved Region 1-alpha protein (E3 CR1-alpha) (Early 3 6.7K protein) (E3-6.7k)
8 entries
Early E3 18.5 kDa glycoprotein (E3-19K) (E3gp 19 kDa) (E19) (GP19K)
1 entry
Pre-early 3 receptor internalization and degradation alpha protein (Pre-E3-RID-alpha protein)
1 entry
Early 3 receptor internalization and degradation beta protein (E3 RID-beta protein) (Early E3B 14.5 kDa protein) (E3-14.5k)
2 entries
Probable early E4 11 kDa protein
1 entry
Probable early E4 13 kDa protein
8 entries
Early E4 34 kDa protein
1 entry
E4-ORF1 (EC 3.6.1.23) (Early E4 14.0 kDa protein) (ORF1) (Probable dUTPase E4 ORF1)
1 entry
Early 4 ORF1 protein (E4-ORF1) (E4 ORF1 control protein)
1 entry
Early 4 ORF2 protein (E4-ORF2)
1 entry
Early 4 ORF3 protein (E4-ORF3) (E4 ORF3 control protein) (Early 4 11 kDa protein) (E4-11k)
1 entry
Early 4 ORF4 protein (E4 ORF4) (Early E4 13 kDa protein)
1 entry
Early 4 ORF6/7 control protein (E4-ORF6/7) (Early E4 17 kDa protein)
6 entries
Late L2 mu core protein (Protein X) (pX) (pMu)
2 entries
I-leader protein
11 entries
Pre-histone-like nucleoprotein (Pre-core protein VII) (pVII)
10 entries
Packaging protein 1 (Packaging protein IVa2)
3 entries
Packaging protein 2 (Packaging protein 22K) (L4-22K)
6 entries
Packaging protein 3 (L1-52/55 kDa protein) (Packaging protein 52K)
19 entries
Protease (EC 3.4.22.39) (Adenain) (Adenovirus protease) (AVP) (Adenovirus proteinase) (Endoprotease)
5 entries
Protein 33K (L4-33K) (Splicing factor 33K) (Terminase, small subunit)
8 entries
Shutoff protein (100 kDa protein) (p100K) (100K-chaperone protein) (L4-100K) (Shutoff protein 100K)
2 entries
Fiber protein 1
2 entries
Fiber protein 2
20 entries
Fiber protein (SPIKE) (Protein IV)
9 entries
Preterminal protein (pTP) (Bellett protein) (Precursor terminal protein)
2 entries
U exon protein (UXP)
1 entry
Uncharacterized 10.4 kDa early protein
1 entry
Uncharacterized protein F-112
1 entry
Uncharacterized 11.3 kDa early protein
2 entries
Uncharacterized 11.5 kDa early protein
1 entry
Uncharacterized 11.6 kDa early protein
1 entry
Uncharacterized protein F-121
1 entry
Uncharacterized 12.6 kDa early protein
1 entry
Uncharacterized protein B-137
1 entry
Uncharacterized 14.5 kDa early protein
1 entry
Uncharacterized protein C-168
1 entry
Uncharacterized protein D-172
1 entry
Uncharacterized 19.0 kDa early protein
1 entry
Uncharacterized 20.6 kDa early protein
1 entry
Uncharacterized protein F-215
1 entry
Uncharacterized protein E-95
1 entry
Uncharacterized 9.7 kDa early protein
1 entry
Uncharacterized 16.5 kDa protein in 100 kDa protein region
1 entry
Uncharacterized 8.0 kDa protein in 100 kDa protein region
1 entry
Uncharacterized 6.9 kDa protein in 100 kDa protein region
1 entry
Uncharacterized 7.3 kDa protein in 100 kDa protein region
1 entry
Uncharacterized 12.4 kDa protein in 33 kDa protein region
Bat adenovirus 2 taxid:696069
Bat adenovirus TJM taxid:727977
Bat mastadenovirus WIV10 taxid:1788432
Bat mastadenovirus WIV11 taxid:1788433
Bat mastadenovirus WIV12 taxid:1788434
Bat mastadenovirus WIV9 taxid:1788436
Canine adenovirus serotype 1 taxid:10512
Canine adenovirus serotype 1 (strain RI261) taxid:69151
Canine mastadenovirus A taxid:10537
Chimpanzee adenovirus Y25 taxid:1123958
Equine adenovirus A serotype 1 taxid:46916
Equine adenovirus B serotype 2 taxid:67603
Human adenovirus 55 taxid:714978
| Protein | ModelArchive |
| L2 (Protein X) | ma-jd-viral-44608 |
| Protease (EC 3.4.22.39) (Adenain) (Adenovirus protease) (AVP) (Adenovirus proteinase)... | ma-jd-viral-13987 |
Human adenovirus A serotype 12 taxid:28282
Human adenovirus C serotype 2 taxid:10515
Human adenovirus C serotype 5 taxid:28285
| Protein | ModelArchive |
| 14.3 kDa protein (Control protein E4orf1) (E4 orf 1) | ma-jd-viral-04366 |
| 34 kDa protein (E4 34K) (E4 orf 6) | ma-jd-viral-05105 |
