Modulation of host E3 ubiquitin ligases by virus (kw:KW-1123)
Numerous cellular processes are regulated by the reversible conjugation of ubiquitin (UB) or ubiquitin-like (UBL) proteins to substrates. The E3 ubiquitin ligase operates in conjunction with an E1 ubiquitin-activating enzyme and an E2 ubiquitin-conjugating enzyme and transfers ubiquitin to the target substrate. Two types of E3 ubiquitin ligases are present in cells based on the structure of their catalytic domain named RING and HECT. The E3s polyubiquitinate target proteins to a cellular organelle known as the 26S proteasome where substrates are degraded into small peptides. Proteasomal degradation is critical component of many cellular processes including cell cycle regulation,induction of the inflammatory response and antigen presentation.
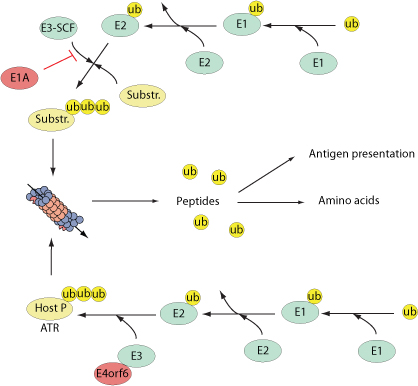
Several viruses encode viral E3 ligases in order to direct specific substrates for proteasomal degradation. For example, herpes simplex virus encodes ICPO which acts as a nuclear E3 ubiquitin ligase that polyubiquitinates the nuclear bodies-associated PML and Sp100 proteins.
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)26 entries grouped by protein
7 entries
Early E1A protein (Early E1A 27 kDa protein)
7 entries
Kelch repeat and BTB domain-containing protein 1
4 entries
Kelch repeat and BTB domain-containing protein 2
3 entries
Latent membrane protein 2 (Terminal protein)
1 entry
Large T antigen (LT) (LT-AG) (EC 5.6.2.4) (DNA 3'-5' helicase large T antigen)
1 entry
Ankyrin repeat domain-containing protein M-T5 (M-T5 protein) (M5)
3 entries
