Inhibition of host MAVS by virus (kw:KW-1097)
The MAVS protein is required for innate immune defense against viruses. It acts downstream of RIG-I and MDA5, which detect intracellular dsRNA produced during viral replication, to coordinate pathways leading to induction of antiviral cytokines. Alternatively, MAVS can cause apoptosis independent of its function in initiating interferon production. MAVS harbors a C-terminal transmembrane domain that targets it to the mitochondrial outer membrane.
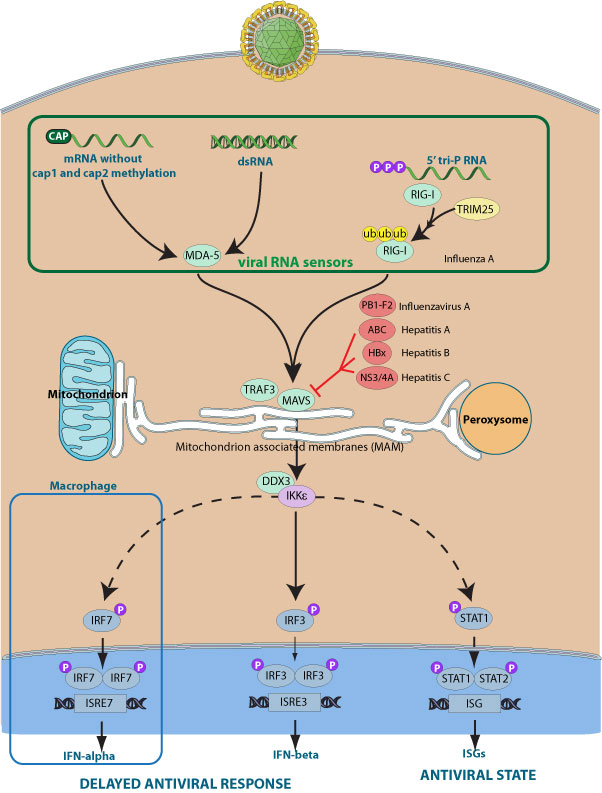
Some viruses selectively inhibit MAVS function. For example, Hepatitis C virus encodes a specific serine protease NS3/4A which cleaves the MAVS transmembrane domain, removes it from the mitochondria thereby preventing interferon production. In a similar way, Hepatitis A encodes for the ABC protein, which localizes to the mitochondria and inhibits MAVS signaling via proteolytic cleavage.
Zsuzsanna T Varga, Alesha Grant, Balaji Manicassamy, Peter Palese
J. Virol. August 2012; 86: 8359?8366
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)210 entries grouped by strain
2 entries
Bovine respiratory syncytial virus (strain A51908) (BRS) reference strain
2 entries
Human respiratory syncytial virus B (strain B1) reference strain
2 entries
Influenza A virus (strain A/Puerto Rico/8/1934 H1N1) reference strain
1 entry
Andes orthohantavirus (ANDV) (Andes virus) reference strain
1 entry
Dengue virus type 1 (strain Nauru/West Pac/1974) (DENV-1) reference strain
1 entry
Dobrava-Belgrade orthohantavirus (DOBV) (Dobrava virus) reference strain
1 entry
Hantaan virus (strain 76-118) (Korean hemorrhagic fever virus) reference strain
1 entry
Hepatitis C virus genotype 1a (isolate H77) (HCV) reference strain
1 entry
Hepatitis GB virus B (GBV-B) (GB virus B) reference strain
1 entry
Human hepatitis A virus genotype IB (isolate HM175) (HHAV) (Human hepatitis A virus (isolate Human/Australia/HM175/1976)) reference strain
1 entry
Human metapneumovirus (strain CAN97-83) (HMPV) reference strain
1 entry
Influenza A virus (strain A/Goose/Guangdong/1/1996 H5N1 genotype Gs/Gd) reference strain
1 entry
Poliovirus type 1 (strain Mahoney) reference strain
1 entry
Seneca Valley virus (isolate -/United States/SSV-001/2002) (SVV) reference strain
1 entry
Severe acute respiratory syndrome coronavirus (SARS-CoV) reference strain
1 entry
Severe acute respiratory syndrome coronavirus 2 (2019-nCoV) (SARS-CoV-2) reference strain
1 entry
Sin Nombre orthohantavirus (SNV) (Sin Nombre virus) reference strain
1 entry
Tembusu virus (TMUV) reference strain
1 entry
Tula orthohantavirus (TULV) (Tula virus) reference strain
1 entry
Uukuniemi virus (strain S23) (UUKV) reference strain
2 entries
Human respiratory syncytial virus
2 entries
Human respiratory syncytial virus A (strain A2)
2 entries
Human respiratory syncytial virus B (strain 18537)
2 entries
Influenza A virus (strain A/Ann Arbor/6/1960 H2N2)
2 entries
Influenza A virus (strain A/Beijing/39/1975 H3N2)
2 entries
Influenza A virus (strain A/Brevig Mission/1/1918 H1N1) (Influenza A virus (strain A/South Carolina/1/1918 H1N1))
2 entries
Influenza A virus (strain A/Duck/Germany/1949 H10N7)
2 entries
Influenza A virus (strain A/Henry/1936 H1N1)
2 entries
Influenza A virus (strain A/Hickox/1940 H1N1)
2 entries
Influenza A virus (strain A/Hong Kong/1/1968 H3N2)
2 entries
Influenza A virus (strain A/Hong Kong/5/1983 H3N2)
2 entries
Influenza A virus (strain A/Memphis/1/1971 H3N2)
2 entries
Influenza A virus (strain A/Memphis/101/1972 H3N2)
2 entries
Influenza A virus (strain A/Memphis/102/1972 H3N2)
2 entries
Influenza A virus (strain A/Memphis/110/1976 H3N2)
2 entries
Influenza A virus (strain A/Memphis/18/1978 H3N2)
2 entries
Influenza A virus (strain A/Memphis/2/1978 H3N2)
2 entries
Influenza A virus (strain A/Memphis/4/1980 H3N2)
2 entries
Influenza A virus (strain A/Port Chalmers/1/1973 H3N2)
2 entries
Influenza A virus (strain A/Russia:St.Petersburg/8/2006 H1N1)
2 entries
Influenza A virus (strain A/Swine/Colorado/1/1977 H3N2)
2 entries
Influenza A virus (strain A/USA:Huston/AA/1945 H1N1)
2 entries
Influenza A virus (strain A/USA:Iowa/1943 H1N1)
2 entries
Influenza A virus (strain A/USA:Phila/1935 H1N1)
2 entries
Influenza A virus (strain A/Wilson-Smith/1933 H1N1) (Influenza A virus (strain A/WS/1933 H1N1))
1 entry
Black Creek Canal orthohantavirus (BCCV) (Black Creek Canal virus)
1 entry
Bovine respiratory syncytial virus (strain 391-2) (BRS)
1 entry
Bovine respiratory syncytial virus (strain Rb94) (BRS)
1 entry
Coxsackievirus B3 (strain Nancy)
1 entry
Coxsackievirus B3 (strain Woodruff)
1 entry
Dengue virus type 1 (strain Brazil/97-11/1997) (DENV-1)
1 entry
Dengue virus type 1 (strain Singapore/S275/1990) (DENV-1)
1 entry
Dengue virus type 2 (isolate Thailand/0168/1979) (DENV-2)
1 entry
Dengue virus type 2 (strain 16681-PDK53) (DENV-2)
1 entry
Dengue virus type 2 (strain Jamaica/1409/1983) (DENV-2)
1 entry
Dengue virus type 2 (strain Peru/IQT2913/1996) (DENV-2)
1 entry
Dengue virus type 2 (strain Puerto Rico/PR159-S1/1969) (DENV-2)
1 entry
Dengue virus type 2 (strain Thailand/16681/1984) (DENV-2)
1 entry
Dengue virus type 2 (strain Thailand/NGS-C/1944) (DENV-2)
1 entry
Dengue virus type 3 (strain China/80-2/1980) (DENV-3)
1 entry
Dengue virus type 3 (strain Martinique/1243/1999) (DENV-3)
1 entry
Dengue virus type 3 (strain Philippines/H87/1956) (DENV-3)
1 entry
Dengue virus type 3 (strain Singapore/8120/1995) (DENV-3)
1 entry
Dengue virus type 3 (strain Sri Lanka/1266/2000) (DENV-3)
1 entry
Dengue virus type 4 (strain Dominica/814669/1981) (DENV-4)
1 entry
Dengue virus type 4 (strain Philippines/H241/1956) (DENV-4)
1 entry
Dengue virus type 4 (strain Singapore/8976/1995) (DENV-4)
1 entry
Dengue virus type 4 (strain Thailand/0348/1991) (DENV-4)
1 entry
Dengue virus type 4 (strain Thailand/0476/1997) (DENV-4)
1 entry
Hepatitis C virus genotype 1a (isolate 1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate BK) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Con1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HC-J1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HC-JT) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HCR6) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Japanese) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Taiwan) (HCV)
1 entry
Hepatitis C virus genotype 1b (strain HC-J4) (HCV)
1 entry
Hepatitis C virus genotype 1c (isolate HC-G9) (HCV)
1 entry
Hepatitis C virus genotype 1c (isolate India) (HCV)
1 entry
Hepatitis C virus genotype 2a (isolate HC-J6) (HCV)
1 entry
Hepatitis C virus genotype 2a (isolate JFH-1) (HCV)
1 entry
Hepatitis C virus genotype 2b (isolate HC-J8) (HCV)
1 entry
Hepatitis C virus genotype 2b (isolate JPUT971017) (HCV)
1 entry
Hepatitis C virus genotype 2c (isolate BEBE1) (HCV)
1 entry
Hepatitis C virus genotype 2k (isolate VAT96) (HCV)
1 entry
Hepatitis C virus genotype 3a (isolate NZL1) (HCV)
1 entry
Hepatitis C virus genotype 3a (isolate k3a) (HCV)
1 entry
Hepatitis C virus genotype 3b (isolate Tr-Kj) (HCV)
1 entry
Hepatitis C virus genotype 3k (isolate JK049) (HCV)
1 entry
Hepatitis C virus genotype 4a (isolate ED43) (HCV)
1 entry
Hepatitis C virus genotype 5a (isolate EUH1480) (HCV)
1 entry
Hepatitis C virus genotype 5a (isolate SA13) (HCV)
1 entry
Hepatitis C virus genotype 6a (isolate 6a33) (HCV)
1 entry
Hepatitis C virus genotype 6a (isolate EUHK2) (HCV)
1 entry
Hepatitis C virus genotype 6b (isolate Th580) (HCV)
1 entry
Hepatitis C virus genotype 6d (isolate VN235) (HCV)
1 entry
Hepatitis C virus genotype 6g (isolate JK046) (HCV)
1 entry
Hepatitis C virus genotype 6h (isolate VN004) (HCV)
1 entry
Hepatitis C virus genotype 6k (isolate VN405) (HCV)
1 entry
Human enterovirus 71 (EV71) (EV-71)
1 entry
Human enterovirus 71 (strain 7423/MS/87) (EV71) (EV-71)
1 entry
Human enterovirus 71 (strain USA/BrCr/1970) (EV71) (EV-71)
1 entry
Human hepatitis A virus genotype IA (isolate GBM) (HHAV) (Human hepatitis A virus (isolate Human/Germany/GBM/1976))
1 entry
Human hepatitis A virus genotype IA (isolate H2) (HHAV) (Human hepatitis A virus (isolate Human/China/H2/1982))
1 entry
Human hepatitis A virus genotype IA (isolate LA) (HHAV) (Human hepatitis A virus (isolate Human/Northern California/LA/1974))
1 entry
Human hepatitis A virus genotype IB (isolate MBB) (HHAV) (Human hepatitis A virus (isolate Human/Northern Africa/MBB/1978))
1 entry
Human hepatitis A virus genotype IIA (isolate CF-53) (HHAV) (Human hepatitis A virus (isolate Human/France/CF-53/1979))
1 entry
Human hepatitis A virus genotype IIB (isolate SLF88) (HHAV) (Human hepatitis A virus (isolate Human/Sierra Leone/SLF88/1988))
1 entry
Human hepatitis A virus genotype IIIA (isolate NOR-21) (HHAV) (Human hepatitis A virus (isolate Human/Norway/NOR-21/1998))
1 entry
Human hepatitis A virus genotype IIIB (isolate HAJ85-1) (HHAV) (Human hepatitis A virus (isolate Human/Japan/HAJ85-1/1985))
1 entry
Human respiratory syncytial virus A (strain S-2) (HRSV-S2)
1 entry
Influenza A virus (strain A/Brazil/11/1978 H1N1)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/31.2/2002 H5N1 genotype X1)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/37.4/2002 H5N1 genotype X2)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/715.5/2001 H5N1 genotype E)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/96.1/2002 H5N1 genotype Y)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/FY150/2001 H5N1 genotype D)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/YU22/2002 H5N1 genotype Z)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/YU562/2001 H5N1 genotype B)
1 entry
Influenza A virus (strain A/Chicken/Pennsylvania/1/1983 H5N2)
1 entry
Influenza A virus (strain A/Chicken/Pennsylvania/1370/1983 H5N2)
1 entry
Influenza A virus (strain A/Chicken/Scotland/1959 H5N1)
1 entry
Influenza A virus (strain A/Chicken/Shantou/4231/2003 H5N1 genotype V)
1 entry
Influenza A virus (strain A/Chicken/Victoria/1/1985 H7N7)
1 entry
Influenza A virus (strain A/Chile/1/1983 H1N1)
1 entry
Influenza A virus (strain A/China:Nanchang/11/1996 H1N1)
1 entry
Influenza A virus (strain A/Duck/Czechoslovakia/1956 H4N6)
1 entry
Influenza A virus (strain A/Duck/England/1/1956 H11N6)
1 entry
Influenza A virus (strain A/Duck/Hokkaido/8/1980 H3N8)
1 entry
Influenza A virus (strain A/Duck/Hong Kong/2986.1/2000 H5N1 genotype C)
1 entry
Influenza A virus (strain A/England/878/1969 H3N2)
1 entry
Influenza A virus (strain A/Fort Monmouth/1/1947 H1N1)
1 entry
Influenza A virus (strain A/Goose/Guangxi/345/2005 H5N1 genotype G)
1 entry
Influenza A virus (strain A/Grey teal/Australia/2/1979 H4N4)
1 entry
Influenza A virus (strain A/Guinea fowl/Hong Kong/38/2002 H5N1 genotype X0)
1 entry
Influenza A virus (strain A/Gull/Maryland/704/1977 H13N6)
1 entry
Influenza A virus (strain A/Hong Kong/156/1997 H5N1 genotype Gs/Gd)
1 entry
Influenza A virus (strain A/Hong Kong/212/2003 H5N1 genotype Z+)
1 entry
Influenza A virus (strain A/India/6263/1980 H1N1)
1 entry
Influenza A virus (strain A/Japan/305/1957 H2N2)
1 entry
Influenza A virus (strain A/Kiev/59/1979 H1N1)
1 entry
Influenza A virus (strain A/Kitakyushu/159/1993 H3N2)
1 entry
Influenza A virus (strain A/Korea/426/1968 H2N2)
1 entry
Influenza A virus (strain A/Leningrad/134/17/1957 H2N2)
1 entry
Influenza A virus (strain A/Leningrad/134/1957 H2N2)
1 entry
Influenza A virus (strain A/Leningrad/134/47/1957 H2N2)
1 entry
Influenza A virus (strain A/Malaysia:Malaya/302/1954 H1N1)
1 entry
Influenza A virus (strain A/Memphis/8/1988 H3N2)
1 entry
Influenza A virus (strain A/New Zealand:South Canterbury/35/2000 H1N1)
1 entry
Influenza A virus (strain A/Northern Territory/60/1968 H3N2) (Influenza A virus (strain NT60)) (Influenza A virus (strain A/NT/60/1968 H3N2))
1 entry
Influenza A virus (strain A/Qu/7/1970 H3N2)
1 entry
Influenza A virus (strain A/Seal/Massachusetts/1/1980 H7N7)
1 entry
Influenza A virus (strain A/Silky Chicken/Hong Kong/SF189/2001 H5N1 genotype A)
1 entry
Influenza A virus (strain A/Silky Chicken/Hong Kong/YU100/2002 H5N1 genotype X3)
1 entry
Influenza A virus (strain A/Singapore/1/1957 H2N2)
1 entry
Influenza A virus (strain A/Swine/Iowa/15/1930 H1N1)
1 entry
Influenza A virus (strain A/Swine/Tennessee/24/1977 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1961 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1967 H1N1)
1 entry
Influenza A virus (strain A/Teal/China/2978.1/2002 H5N1 genotype W)
1 entry
Influenza A virus (strain A/Tokyo/3/1967 H2N2)
1 entry
Influenza A virus (strain A/Turkey/Ireland/1378/1983 H5N8)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/501/1978 H6N8)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/833/1980 H4N2)
1 entry
Influenza A virus (strain A/Turkey/Ontario/6118/1968 H8N4)
1 entry
Influenza A virus (strain A/Turkey/Ontario/7732/1966 H5N9)
1 entry
Influenza A virus (strain A/Turkey/Wisconsin/1/1966 H9N2)
1 entry
Influenza A virus (strain A/USA:Albany/12/1951 H1N1)
1 entry
Influenza A virus (strain A/USA:Memphis/10/1996 H1N1)
1 entry
Influenza A virus (strain A/USA:Texas/UR06-0195/2007 H1N1)
1 entry
Influenza A virus (strain A/USSR/90/1977 H1N1)
1 entry
Influenza A virus (strain A/Udorn/307/1972 H3N2)
1 entry
Influenza A virus (strain A/Victoria/3/1975 H3N2)
1 entry
Influenza A virus (strain A/X-31 H3N2)
1 entry
Ovine respiratory syncytial virus (strain WSU 83-1578) (ORSV)
1 entry
Poliovirus type 1 (strain Sabin)
1 entry
Poliovirus type 2 (strain Lansing)
1 entry
Poliovirus type 2 (strain W-2)
1 entry
Poliovirus type 3 (strain 23127)
1 entry
Poliovirus type 3 (strains P3/Leon/37 and P3/Leon 12A[1]B)
1 entry
