Hepadnaviridae (taxid:10404)
VIRION
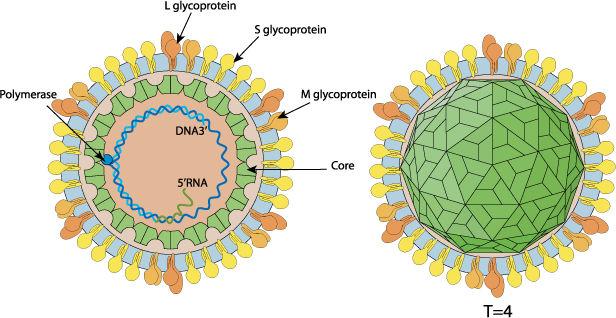
Enveloped, spherical. Diameter from about 42nm. Icosahedric capsid with a T=4 symmetry.
GENOME
Partially dsDNA circular genome, about 3.2 kb in size. Encodes for 7 proteins. On rare non-specific recombination, the viral genome can be integrated in host chromosome. This inactivates the integrated virus but can gives the host cell a replicative advantage sometimes leading to hepatocarcinoma.
GENE EXPRESSION
The minichromosome is transcribed by cellular RNA polymerase II under the control of three to four promoters (the core, pre-S1, pre-S2/S promoters, and the X promoter in mammalian viruses) and two enhancer regions (ENH1 and ENH2). The pre-genomic RNA is alternatively spliced. The unspliced form is exported from the nucleus through a PRE motif possibly by capsid protein. The Polymerase and short S proteins are expressed by leaky scanning from the pg mRNA and the S mRNA respectively.
ENZYMES
- Reverse transcriptase
- RNAse H [P]
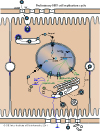
REPLICATION
CYTOPLASMIC/NUCLEAR
- Virus attaches to host receptors through major surface antigen and enters the cell by an unknown mechanism.
- Relaxed circular DNA (RC-DNA) and capsid are transported via microtubules to the nucleus where DNA is released through the nuclear pore, and repaired to form covalently closed circular DNA (cccDNA).
- Transcription by RNA polymerase II of the pregenomic RNA (pgRNA) and subgenomic mRNAs, inducing synthesis of all the viral proteins.
- pgRNA is encapsidated, together with the P protein, and reverse-transcribed inside the nucleocapsid in (-)DNA covalently linked to P protein.
- (+)DNA synthesis from the (-)DNA template generates new RC-DNA.
- Transport to the nucleus of new RC-DNA leads to cccDNA amplification; alternatively, the RC-DNA containing nucleocapsids are enveloped at the ER, and new virions are released by exocytosis.
Host-virus interaction
Primary receptor
HBV L surface protein can bind to heparan sulfate
Entry receptor
HBV L surface protein Binds to SLC10A1/NTCP  in order to enter hepatocytes.
in order to enter hepatocytes.
Autophagy modulation
HBV protein HBx seems to be able to increase autophagy through the up-regulation of host beclin 1 expression  .
.
Cell-cycle modulation
HBV protein HBx protein deregulates cell cycle checkpoint controls
at G0/G1 and G2/M  .
.
Innate immune response inhibition
HBV protein HBx directly interacts with and inhibits host MAVS protein by inducing its ubiquitination and targeting it for degradation  .
.
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)292 entries grouped by protein
56 entries
Capsid protein (Core antigen) (Core protein) (HBcAg) (p21.5)
65 entries
Protein P
49 entries
External core antigen (HBeAg) (Precore protein) (p25)
64 entries
Large envelope protein (L glycoprotein) (L-HBsAg) (LHB) (Large S protein) (Large surface protein) (Major surface antigen)
55 entries
Protein X (HBx) (Peptide X) (pX)
1 entry
Putative X-Core fused protein
1 entry
Putative uncharacterized 10.4 kDa protein
1 entry
Putative uncharacterized 15.3 kDa protein
 (ModelArchive).
(ModelArchive).
Hepatitis B virus taxid:10407
Hepatitis B virus genotype D subtype ayw (isolate France/Tiollais/1979) taxid:490133
| Protein | ModelArchive |
| Capsid protein (Core antigen) (Core protein) (HBcAg) (p21.5) | ma-jd-viral-16247 |
Horseshoe bat hepatitis B virus taxid:1508711
Parrot hepatitis B virus taxid:1128118
Roundleaf bat hepatitis B virus taxid:1508710
Sheldgoose hepatitis B virus taxid:259898
Snow goose hepatitis B virus taxid:89623
Tent-making bat hepatitis B virus taxid:1508712
| Protein | ModelArchive |
| Core protein | ma-jd-viral-16240 |
| Protein P (EC 2.7.7.49) (EC 2.7.7.7) | ma-jd-viral-49618 |
| Protein X (HBx) (Peptide X) (pX) | ma-jd-viral-29348 |
| Surface protein | ma-jd-viral-48178 |
White sucker hepatitis B virus taxid:1690672
| Protein | ModelArchive |
| Large envelope protein | ma-jd-viral-48096 |
| PreC/core protein | ma-jd-viral-16238 |
| Protein P (EC 2.7.7.49) (EC 2.7.7.7) (EC 3.1.26.4) | ma-jd-viral-49619 |