Rhadinovirus (taxid:10379)
VIRION
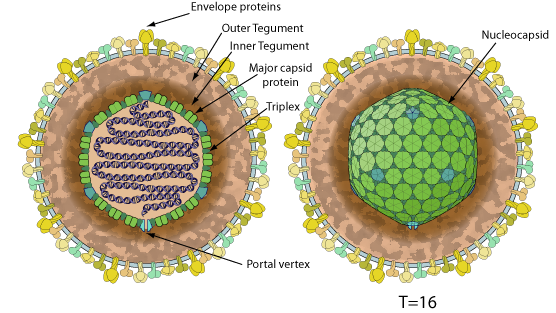
Enveloped, spherical to pleomorphic, 150-200 nm in diameter, T=16 icosahedral symmetry. Capsid consists of 162 capsomers and is surrounded by an amorphous tegument. Glycoproteins complexes are embedded in the lipid envelope.
GENOME
Monopartite, linear, dsDNA genome of about 180 kb. The genome contains terminal and internal reiterated sequences.
GENE EXPRESSION
Each viral transcript usually encodes a single protein and has a promoter/regulatory sequence, a TATA box, a transcription initiation site, a 5' leader sequence of 30-300 bp (not translated), a 3' untranslated sequence of 10-30 bp, and a poly A signal. There are many gene overlaps. There are only few spliced genes. Some of the expressed ORFs are antisense to each other. Some ORFs can be accessed from more than one promoter. Certain proteins are downregulated translationaly by a leaky scanning from an upstream ORF.
ENZYMES
- DNA-dependent DNA polymerase
- DNA primase
- Tegument deneddylase (Peptidase C76)
- Assemblin (Peptidase S21)
- Kinase
- Helicase
- Ribonucleoside-diphosphate reductase
- Thymidine kinase
- Uracil-DNA glycosylase
REPLICATION
NUCLEAR
Lytic replication:
- Attachment of the viral glycoproteins to host receptors mediates endocytosis of the virus into the host cell.
- Fusion with the plasma membrane to release the core and the tegument proteins into the host cytoplasm.
- The capsid is transported to the nuclear pore where the viral DNA is released into the nucleus.
- Transcription of immediate early genes which promote transcription of early genes and protect the virus against innate host immunity.
- Transcription of early viral mRNA by host polymerase II, encoding proteins involved in replication of the viral DNA.
- A first round of circular genome amplification occurs by bidirectional replication
- Synthesis of linear concatemer copies of viral DNA by rolling circle.
- Transcription of late mRNAs by host polymerase II, encoding structural proteins.
- Assembly of the virus in nuclear viral factories and budding through the inner lamella of the nuclear membrane which has been modified by the insertion of herpes glycoproteins, throughout the Golgi and final release at the plasma membrane.
Latent replication : replication of circular viral episome in tandem with the host cell DNA using the host cell replication machinery.
Host-virus interaction
Adaptive immune response inhibition
Kaposi proteins MIR1/K3 and MIR2/K5 are E3 ubiquitin ligase which promote the ubiquitination and subsequent degradation of host MHC-I and CD1D molecules, thereby inhibiting the host adaptive immune response.
Apoptosis modulation
Kaposi sarcoma-associated herpesvirus encodes a viral FLICE inhibitory protein (vFLIP) that blocks apoptosis  .
.
Autophagy modulation
The KSHV v-Bcl-2 inhibits host autophagy by interacting with host beclin-1  .
.
Cell-cycle modulation
KSHV v-cyclin encoded by ORF72, is a cyclin-like protein that shares homology to cellular D-type cyclins. Like human cyclins, v-cyclin interacts with cdk4 and cdk6 to activate these kinases and promote the G1/S phase transition.
The conserved UL24 family of human alpha, beta and gamma herpesviruses induces a cell cycle arrest at G2/M transition through inactivation of the host cyclinB/cdc2 complex. EBV encodes an UL24 homolog termed ORF20 that should perform this role  .
.
Innate immune response inhibition
Human herpes virus 8 (HHV-8) has developed unique mechanisms to modulate host immune system by incorporating viral homologs to several cellular regulatory genes into its genome. For instance, vIRF-1 is able to bind to both IRF-3 and IRF-7 and efficiently inhibits the formation of transcriptionally competent IRF-3-CBP/p300 complexes  .
.
Splicing inhibition
KSHV ORF57 modulates the viral and host mRNA expression by exporting unspliced mRNA, thereby inducing alternative splicing  .
.
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)181 entries grouped by protein
2 entries
Shutoff alkaline exonuclease (SOX) (EC 3.1.-.-)
2 entries
Apoptosis regulator Bcl-2 homolog
2 entries
Putative transcription activator BRLF1 homolog
1 entry
Complement control protein homolog (CCPH)
1 entry
Surface glycoprotein CD59 homolog
2 entries
Cytoplasmic envelopment protein 1
2 entries
Cytoplasmic envelopment protein 2
2 entries
Cytoplasmic envelopment protein 3
1 entry
Cyclin homolog (V-cyclin)
2 entries
Capsid vertex component 1
2 entries
Capsid vertex component 2
2 entries
Major DNA-binding protein
2 entries
DNA polymerase catalytic subunit (EC 2.7.7.7)
2 entries
Deoxyuridine 5'-triphosphate nucleotidohydrolase (dUTPase) (EC 3.6.1.23) (dUTP pyrophosphatase)
4 entries
Viral dihydrofolate reductase (vDHFR) (EC 1.5.1.3)
2 entries
Envelope glycoprotein B (gB)
4 entries
Beta-1,3-galactosyl-O-glycosyl-glycoprotein beta-1,6-N-acetylglucosaminyltransferase (EC 2.4.1.102) (EC 2.4.1.148) (EC 2.4.1.150) (C2GnT-mucin type) (C2GnT-M)
1 entry
Probable ganciclovir kinase (EC 2.7.1.-)
2 entries
Envelope glycoprotein H (gH)
2 entries
Envelope glycoprotein L (gL)
2 entries
Envelope glycoprotein M (gM)
2 entries
Envelope glycoprotein N
2 entries
DNA replication helicase (EC 3.6.4.-)
1 entry
DNA helicase/primase complex-associated protein (HEPA) (Primase-associated factor)
1 entry
Transcriptional regulator ICP22 homolog (Immediate-early protein)
1 entry
mRNA export factor ICP27 homolog (52 kDa immediate-early phosphoprotein) (EB2 protein homolog)
2 entries
Viral interleukin-17 (vIL-17) (Immediate early gene 13 protein)
2 entries
Inner tegument protein
1 entry
Protein K1
1 entry
Protein K12
1 entry
Protein K15
1 entry
Protein K4.2
1 entry
Protein K6
1 entry
Protein K7
1 entry
Protein K8.1
1 entry
E3 SUMO-protein ligase K-bZIP (EC 2.3.2.-) (E3 SUMO-protein transferase K-bZIP)
2 entries
Thymidine kinase (EC 2.7.1.21)
1 entry
Protein LANA1
3 entries
Large tegument protein deneddylase (EC 3.4.19.12) (EC 3.4.22.-)
2 entries
Major capsid protein (MCP)
2 entries
E3 ubiquitin-protein ligase MIR1 (EC 2.3.2.36) (IE1B protein) (Modulator of immune recognition 1) (ORF K3) (RING-type E3 ubiquitin transferase MIR1)
1 entry
E3 ubiquitin-protein ligase MIR2 (EC 2.3.2.27) (IE1A protein) (Modulator of immune recognition 2) (ORF K5) (RING-type E3 ubiquitin transferase MIR2)
1 entry
Post-transcriptional regulator MTA (MTA) (EB2 protein homolog)
2 entries
Nuclear egress protein 1
2 entries
Nuclear egress protein 2
1 entry
Protein ORF10
1 entry
Protein ORF11
1 entry
Protein ORF23
1 entry
Protein ORF24
1 entry
Protein ORF27
1 entry
Protein ORF28
1 entry
Protein ORF30
1 entry
Protein ORF35
1 entry
Complement control protein (KCP)
1 entry
Protein ORF45
1 entry
Tegument protein ORF48
1 entry
Protein ORF49
1 entry
Replication and transcription activator (RTA) (EC 2.3.2.27)
1 entry
Tegument protein ORF52 (Kaposi's sarcoma-associated herpesvirus inhibitor of cGAS) (KicGAS)
1 entry
Protein ORF58
1 entry
Protein ORF75
1 entry
OX-2 membrane glycoprotein homolog (Viral OX2) (vOX2)
1 entry
DNA polymerase processivity factor
2 entries
Portal protein
2 entries
DNA primase (EC 2.7.7.-)
2 entries
Ribonucleoside-diphosphate reductase large subunit (R1) (EC 1.17.4.1) (Ribonucleotide reductase large subunit)
2 entries
Ribonucleoside-diphosphate reductase small subunit (EC 1.17.4.1) (Ribonucleotide reductase small subunit)
2 entries
Capsid scaffolding protein (Capsid protein P40) (Protease precursor) (pPR)
2 entries
Small capsomere-interacting protein
2 entries
Saimiri transformation-associated protein (Collagen-like protein) (stpC)
2 entries
Gene 55 protein
1 entry
Protein tio
3 entries
Tyrosine-protein kinase-interacting protein (Tip)
2 entries
Tripartite terminase subunit 1
1 entry
Tripartite terminase subunit 2
2 entries
Tripartite terminase subunit 3 (EC 3.1.-.-) (Terminase large subunit)
2 entries
Triplex capsid protein 1
2 entries
Triplex capsid protein 2
1 entry
Transforming protein STP
3 entries
Thymidylate synthase (TS) (TSase) (EC 2.1.1.45)
2 entries
Protein UL24 homolog
2 entries
Packaging protein UL32
2 entries
Protein ORF66
2 entries
Gene 18 protein
1 entry
Gene 24 protein
1 entry
Uncharacterized gene 30 protein
2 entries
Gene 31 protein
2 entries
Gene 34 protein
1 entry
Gene 35 protein
2 entries
Uracil-DNA glycosylase (UDG) (EC 3.2.2.27) (UNG)
1 entry
Protein vCCL3
1 entry
viral cyclin homolog (v-cyclin)
1 entry
Viral FLICE protein (vFLIP)
1 entry
Uncharacterized gene 5 protein
1 entry
Uncharacterized gene 10 protein
1 entry
Uncharacterized gene 11 protein
1 entry
Uncharacterized gene 12 protein
1 entry
Bcl-2-like gene 16 protein
1 entry
Uncharacterized gene 23 protein
1 entry
Uncharacterized gene 27 protein
1 entry
Uncharacterized gene 28 protein
1 entry
Uncharacterized gene 40 protein
1 entry
Uncharacterized gene 41 protein
1 entry
Uncharacterized gene 45 protein
1 entry
Uncharacterized gene 48 protein
1 entry
Uncharacterized gene 49 protein
1 entry
Gene 51 glycoprotein
1 entry
Uncharacterized gene 52 protein
1 entry
Gene 58 protein
1 entry
Gene 59 protein
1 entry
Uncharacterized gene 71 protein
1 entry
G-protein coupled receptor homolog ECRF3
1 entry
viral G-protein coupled receptor (vGPCR) (Protein ORF74)
1 entry
Probable E3 ubiquitin-protein ligase IE1 (EC 2.3.2.27) (32.7 kDa immediate early protein IE1) (RING-type E3 ubiquitin transferase IE1)
1 entry
Immediate-early protein IE-G
1 entry
Viral interleukin-6 homolog (vIL-6)
1 entry
VIRF-1
1 entry
Viral IRF2-like protein (vIRF-2)
1 entry
Viral IRF3-like protein (VIRF-3)
1 entry
Viral IRF4-like protein (vIRF-4)
1 entry
Viral macrophage inflammatory protein 2 (Viral macrophage inflammatory protein II) (vMIP-II) (vMIP-1B)
1 entry
Probable membrane antigen 3 (Tegument protein)
1 entry
Probable membrane antigen 75 (Tegument protein)
1 entry
Viral protein kinase (EC 2.7.11.1)
1 entry
Uncharacterized 9.5 kDa protein in DHFR 3'region (ORF3)
1 entry
