Fusion of virus membrane with host endosomal membrane (kw:KW-1170)
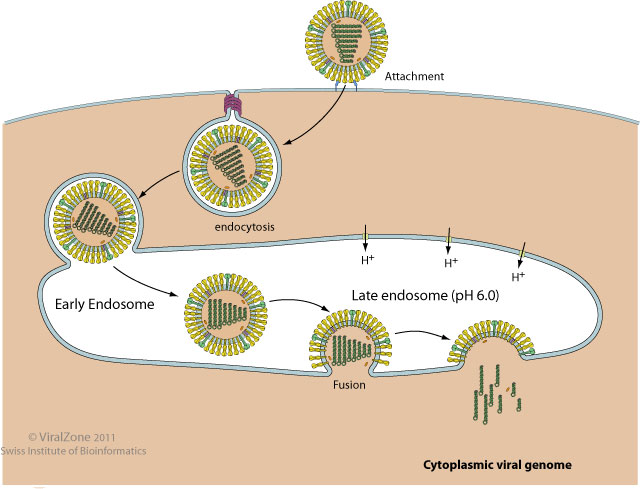
Enveloped viruses use specialized machinery to fuse viral and host cell membranes after internalization through the endosomal pathway.
Viral fusion proteins drive this fusion reaction by undergoing major conformational change. The specific trigger depends on the virus and very often involves the exposure to the low pH of the late endosome.
All fusion proteins are active in a trimeric conformation. Most of them are natively trimeric, some of them are dimers on the virion surface but are converted to trimers upon activation  .
.
Fusion proteins have to be produced in an inactive state in order to avoid to trigger fusion in the Golgi or other compartments upon synthesis and transport to the cell surface. Many are activated in the trans.Golgi network or at the cell surface by proteolytic cleavage which reveals the fusion peptide.
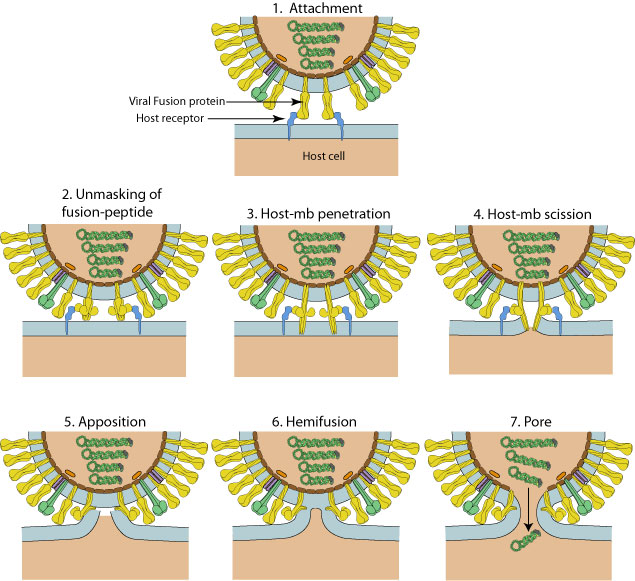
Fusion mechanism according to the "viral fusion pore" model of Lee K.  . A different model has been described with PIV5
. A different model has been described with PIV5
Matching UniProtKB/Swiss-Prot entries
(all links/actions below point to uniprot.org website)538 entries grouped by strain
2 entries
Chikungunya virus (strain S27-African prototype) (CHIKV) reference strain
2 entries
Japanese encephalitis virus (strain Jaoars982) (JEV) reference strain
1 entry
Semliki forest virus (SFV) reference strain
2 entries
Sindbis virus (SINV) reference strain
1 entry
Alethinophid 1 reptarenavirus (isolate AlRrV1/Boa/USA/BC/2009) (Golden Gate virus) reference strain
1 entry
Allpahuayo mammarenavirus (isolate Rat/Peru/CLHP-2472/1997) (ALLV) reference strain
1 entry
Alphavirus salmon subtype 2 (SAV2) (Sleeping disease virus) reference strain
1 entry
Andes orthohantavirus (ANDV) (Andes virus) reference strain
1 entry
Aura virus (AURAV) reference strain
1 entry
Autographa californica nuclear polyhedrosis virus (AcMNPV) reference strain
1 entry
Avian infectious bronchitis virus (strain Beaudette) (IBV) reference strain
1 entry
Banzi virus (BANV) reference strain
1 entry
Barmah forest virus (BFV) reference strain
1 entry
Bat coronavirus HKU4 (BtCoV) (BtCoV/HKU4/2004) reference strain
1 entry
Bat coronavirus HKU5 (BtCoV) (BtCoV/HKU5/2004) reference strain
1 entry
Bat coronavirus HKU9 (BtCoV) (BtCoV/HKU9) reference strain
1 entry
Bear Canyon mammarenavirus (isolate Mouse/United States/AV A0070039/2000) (BCNV) reference strain
1 entry
Bhanja virus (BHAV) reference strain
1 entry
Borna disease virus (strain V) (BDV) reference strain
1 entry
Bovine viral diarrhea virus (isolate NADL) (BVDV) (Mucosal disease virus) reference strain
1 entry
Bovine viral diarrhea virus (strain SD-1) (BVDV) (Mucosal disease virus) reference strain
1 entry
Bunyamwera virus (BUNV) reference strain
1 entry
Bussuquara virus (BUSV) reference strain
1 entry
Chapare mammarenavirus (isolate Human/Bolivia/810419/2003) reference strain
1 entry
Crimean-Congo hemorrhagic fever virus (strain Nigeria/IbAr10200/1970) (CCHFV) reference strain
1 entry
Cupixi mammarenavirus (isolate Rat/Brasil/BeAn 119303/1970) (CPXV) reference strain
1 entry
Dengue virus type 1 (strain Nauru/West Pac/1974) (DENV-1) reference strain
1 entry
Dobrava-Belgrade orthohantavirus (DOBV) (Dobrava virus) reference strain
1 entry
Drosophila melanogaster sigma virus (isolate Drosophila/USA/AP30/2005) (DMelSV) reference strain
1 entry
Dugbe virus (isolate ArD44313) (DUGV) reference strain
1 entry
Eastern equine encephalitis virus (strain PE-3.0815) (EEEV) (Eastern equine encephalomyelitis virus) reference strain
1 entry
Edge Hill virus (EHV) reference strain
1 entry
Getah virus (GETV) reference strain
1 entry
Ground squirrel hepatitis virus (strain 27) (GSHV) reference strain
1 entry
Guanarito mammarenavirus (isolate Human/Venezuela/NH-95551/1990) (GTOV) reference strain
1 entry
Hantaan virus (strain 76-118) (Korean hemorrhagic fever virus) reference strain
1 entry
Hazara virus (isolate JC280) reference strain
1 entry
Hepatitis B virus genotype C subtype ayr (isolate Human/Japan/Okamoto/-) (HBV-C) reference strain
1 entry
Hepatitis B virus genotype D subtype ayw (isolate France/Tiollais/1979) (HBV-D) reference strain
1 entry
Hepatitis C virus genotype 1a (isolate H77) (HCV) reference strain
1 entry
Hepatitis GB virus B (GBV-B) (GB virus B) reference strain
1 entry
Human coronavirus HKU1 (isolate N1) (HCoV-HKU1) reference strain
1 entry
Human coronavirus OC43 (HCoV-OC43) reference strain
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate HXB2) (HIV-1) reference strain
1 entry
Human immunodeficiency virus type 1 group N (isolate YBF30) (HIV-1) reference strain
1 entry
Human immunodeficiency virus type 1 group O (isolate ANT70) (HIV-1) reference strain
1 entry
Human immunodeficiency virus type 2 subtype A (isolate BEN) (HIV-2) reference strain
1 entry
Ilheus virus (ILHV) reference strain
1 entry
Impatiens necrotic spot virus (INSV) reference strain
1 entry
Infectious salmon anemia virus (isolate Atlantic salmon/Norway/810/9/99) (ISAV) reference strain
1 entry
Influenza A virus (strain A/Goose/Guangdong/1/1996 H5N1 genotype Gs/Gd) reference strain
1 entry
Influenza A virus (strain A/Puerto Rico/8/1934 H1N1) reference strain
1 entry
Influenza B virus (strain B/Lee/1940) reference strain
1 entry
Influenza C virus (strain C/Ann Arbor/1/1950) reference strain
1 entry
Ippy mammarenavirus (isolate Rat/Central African Republic/Dak An B 188 d/1970) (IPPYV) reference strain
1 entry
Kokobera virus (KOKV) reference strain
1 entry
Kyasanur forest disease virus (KFDV) reference strain
1 entry
Lake Victoria marburgvirus (strain Musoke-80) (MARV) (Marburg virus (strain Kenya/Musoke/1980)) reference strain
1 entry
Lassa virus (strain Mouse/Sierra Leone/Josiah/1976) (LASV) reference strain
1 entry
Latino mammarenavirus (isolate Rat/Bolivia/MARU 1924/1965) (LATV) reference strain
1 entry
Louping ill virus (LIV) reference strain
1 entry
Lymantria dispar multicapsid nuclear polyhedrosis virus (LdMNPV) reference strain
1 entry
Lymphocytic choriomeningitis virus (strain Armstrong) (LCMV) reference strain
1 entry
Mayaro virus (strain Brazil) (MAYV) reference strain
1 entry
Mobala mammarenavirus (isolate Rat/Central African Republic/Acar 3080/1983) (MOBV) reference strain
1 entry
Murine coronavirus (strain A59) (MHV-A59) (Murine hepatitis virus) reference strain
1 entry
Murray valley encephalitis virus (strain MVE-1-51) (MVEV) reference strain
1 entry
O'nyong-nyong virus (strain SG650) (ONNV) reference strain
1 entry
Oliveros mammarenavirus (isolate Mouse/Argentina/RIID 3229/1990) (OLVV) reference strain
1 entry
Omsk hemorrhagic fever virus (OHFV) reference strain
1 entry
Orgyia pseudotsugata multicapsid polyhedrosis virus (OpMNPV) reference strain
1 entry
Parana mammarenavirus (isolate Rat/Paraguay/12056/1965) (PARV) (Paran mammarenavirus) reference strain
1 entry
Pichinde mammarenavirus (PICV) (Pichind mammarenavirus) reference strain
1 entry
Pirital mammarenavirus (isolate Rat/Venezuela/VAV-488/1995) (PIRV) reference strain
1 entry
Prospect Hill virus (PHV) reference strain
1 entry
Puumala virus (strain Bank vole/Russia/CG1820/1984) reference strain
1 entry
Reston ebolavirus (strain Reston-89) (REBOV) (Reston Ebola virus) reference strain
1 entry
Rift valley fever virus (strain ZH-548 M12) (RVFV) reference strain
1 entry
Rubella virus (strain Therien) (RUBV) reference strain
1 entry
SFTS phlebovirus (isolate SFTSV/Human/China/HB29/2010) (Severe fever with thrombocytopenia virus) reference strain
1 entry
Sabia mammarenavirus (isolate Human/Brasil/SPH114202/1990) (SABV) (Sabi mammarenavirus) reference strain
1 entry
Seoul virus (strain 80-39) reference strain
1 entry
Severe acute respiratory syndrome coronavirus (SARS-CoV) reference strain
1 entry
Severe acute respiratory syndrome coronavirus 2 (2019-nCoV) (SARS-CoV-2) reference strain
1 entry
Sin Nombre orthohantavirus (SNV) (Sin Nombre virus) reference strain
1 entry
Spring viremia of carp virus (Rhabdovirus carpia) reference strain
1 entry
Sudan ebolavirus (strain Human/Uganda/Gulu/2000) (SEBOV) (Sudan Ebola virus) reference strain
1 entry
Tacaribe virus (strain Franze-Fernandez) (TCRV) reference strain
1 entry
Tamiami mammarenavirus (isolate Rat/United States/W 10777/1964) (TAMV) reference strain
1 entry
Tembusu virus (TMUV) reference strain
1 entry
Tibrogargan virus (strain CS132) (TIBV) reference strain
1 entry
Tick-borne encephalitis virus European subtype (strain Neudoerfl) (NEUV) (Neudoerfl virus) reference strain
1 entry
Tick-borne powassan virus (strain LB) (POWV) (Powassan virus) reference strain
1 entry
Tomato spotted wilt virus (strain Brazilian Br-01) (TSWV) reference strain
1 entry
Tula orthohantavirus (TULV) (Tula virus) reference strain
1 entry
Tupaia virus (isolate Tupaia/Thailand/-/1986) (TUPV) reference strain
1 entry
Usutu virus (USUV) reference strain
1 entry
Uukuniemi virus (strain S23) (UUKV) reference strain
1 entry
Venezuelan equine encephalitis virus (strain Trinidad donkey) (VEEV) reference strain
1 entry
Vesicular stomatitis Indiana virus (strain 98COE North America) (VSIV) reference strain
1 entry
Vesicular stomatitis Indiana virus (strain San Juan) (VSIV) reference strain
1 entry
Wesselsbron virus (WSLV) reference strain
1 entry
West Nile virus (WNV) reference strain
1 entry
Whitewater Arroyo mammarenavirus (isolate Rat/United States/AV 9310135/1995) (WWAV) reference strain
1 entry
Woodchuck hepatitis B virus (isolate 7) (WHV) reference strain
1 entry
Woolly monkey hepatitis B virus (isolate Louisville) (WMHBV) reference strain
1 entry
Yellow fever virus (strain 17D vaccine) (YFV) reference strain
1 entry
Zaire ebolavirus (strain Mayinga-76) (ZEBOV) (Zaire Ebola virus) reference strain
1 entry
Zika virus (ZIKV) reference strain
2 entries
Japanese encephalitis virus (strain M28) (JEV)
2 entries
Japanese encephalitis virus (strain SA(v)) (JEV)
2 entries
Japanese encephalitis virus (strain SA-14) (JEV)
1 entry
Semliki forest virus (SFV)
1 entry
Alkhumra hemorrhagic fever virus (ALKV) (Alkhurma hemorrhagic fever virus)
1 entry
Alphavirus salmon subtype 1 (SAV1) (Salmon pancreas disease virus subtype 1)
1 entry
Alphavirus salmon subtype 3 (SAV3) (Norwegian salmonid alphavirus)
1 entry
Avian infectious bronchitis virus (strain 6/82) (IBV)
1 entry
Avian infectious bronchitis virus (strain D274) (IBV)
1 entry
Avian infectious bronchitis virus (strain KB8523) (IBV)
1 entry
Avian infectious bronchitis virus (strain M41) (IBV)
1 entry
Bat coronavirus 133/2005 (BtCoV) (BtCoV/133/2005)
1 entry
Bat coronavirus 279/2005 (BtCoV) (BtCoV/279/2005)
1 entry
Bat coronavirus HKU3 (BtCoV) (SARS-like coronavirus HKU3)
1 entry
Bat coronavirus Rp3/2004 (BtCoV/Rp3/2004) (SARS-like coronavirus Rp3)
1 entry
Black Creek Canal orthohantavirus (BCCV) (Black Creek Canal virus)
1 entry
Borna disease virus 1 (BoDV-1)
1 entry
Bovine Schmallenberg virus (isolate Bovine/BH80/Germany/2011) (SBV)
1 entry
Bovine coronavirus (strain 98TXSF-110-ENT) (BCoV-ENT) (BCV)
1 entry
Bovine coronavirus (strain 98TXSF-110-LUN) (BCoV-LUN) (BCV)
1 entry
Bovine coronavirus (strain F15) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain L9) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain LSU-94LSS-051) (BCoV-LSU) (BCV)
1 entry
Bovine coronavirus (strain LY-138) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain Mebus) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain OK-0514) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain Quebec) (BCoV) (BCV)
1 entry
Bovine coronavirus (strain vaccine) (BCoV) (BCV)
1 entry
Bovine viral diarrhea virus (strain CP7) (BVDV) (Mucosal disease virus)
1 entry
Bunyavirus La Crosse
1 entry
Bunyavirus germiston
1 entry
Bunyavirus snowshoe hare
1 entry
Chikungunya virus (strain 37997) (CHIKV)
1 entry
Chikungunya virus (strain Nagpur) (CHIKV)
1 entry
Chimpanzee hepatitis B virus (isolate United Kingdom/LSH/1988) (HBVcpz)
1 entry
Choristoneura fumiferana nuclear polyhedrosis virus (CfMNPV)
1 entry
Classical swine fever virus (strain Alfort/Tuebingen) (CSFV) (Hog cholera virus)
1 entry
Classical swine fever virus (strain Brescia) (CSFV) (Hog cholera virus)
1 entry
Dabie bandavirus (Severe fever with thrombocytopenia virus) (Huaiyangshan banyangvirus)
1 entry
Dengue virus type 1 (strain Brazil/97-11/1997) (DENV-1)
1 entry
Dengue virus type 1 (strain Singapore/S275/1990) (DENV-1)
1 entry
Dengue virus type 2 (isolate Thailand/0168/1979) (DENV-2)
1 entry
Dengue virus type 2 (strain 16681-PDK53) (DENV-2)
1 entry
Dengue virus type 2 (strain Jamaica/1409/1983) (DENV-2)
1 entry
Dengue virus type 2 (strain Peru/IQT2913/1996) (DENV-2)
1 entry
Dengue virus type 2 (strain Puerto Rico/PR159-S1/1969) (DENV-2)
1 entry
Dengue virus type 2 (strain Thailand/16681/1984) (DENV-2)
1 entry
Dengue virus type 2 (strain Thailand/NGS-C/1944) (DENV-2)
1 entry
Dengue virus type 3 (strain China/80-2/1980) (DENV-3)
1 entry
Dengue virus type 3 (strain Martinique/1243/1999) (DENV-3)
1 entry
Dengue virus type 3 (strain Philippines/H87/1956) (DENV-3)
1 entry
Dengue virus type 3 (strain Singapore/8120/1995) (DENV-3)
1 entry
Dengue virus type 3 (strain Sri Lanka/1266/2000) (DENV-3)
1 entry
Dengue virus type 4 (strain Dominica/814669/1981) (DENV-4)
1 entry
Dengue virus type 4 (strain Philippines/H241/1956) (DENV-4)
1 entry
Dengue virus type 4 (strain Singapore/8976/1995) (DENV-4)
1 entry
Dengue virus type 4 (strain Thailand/0348/1991) (DENV-4)
1 entry
Dengue virus type 4 (strain Thailand/0476/1997) (DENV-4)
1 entry
Eastern equine encephalitis virus (EEEV) (Eastern equine encephalomyelitis virus)
1 entry
Eastern equine encephalitis virus (strain Florida 91-469) (EEEV) (Eastern equine encephalomyelitis virus)
1 entry
Eastern equine encephalitis virus (strain PE-0.0155) (EEEV) (Eastern equine encephalomyelitis virus)
1 entry
Eastern equine encephalitis virus (strain va33[ten broeck]) (EEEV) (Eastern equine encephalomyelitis virus)
1 entry
Gibbon hepatitis B virus subtype ayw3q (isolate Hope) (HBVgbn)
1 entry
Gorilla hepatitis B virus (isolate Cameroon/gor97) (HBVgor)
1 entry
Hantaan virus (strain B-1) (Korean hemorrhagic fever virus)
1 entry
Hantaan virus (strain Hojo) (Hojo virus) (Korean hemorrhagic fever virus)
1 entry
Hantaan virus (strain Lee) (Lee virus) (Korean hemorrhagic fever virus)
1 entry
Heartland virus (HTRV)
1 entry
Hepatitis B virus genotype A1 subtype adw (isolate Philippines/pFDW294/1988) (HBV-A)
1 entry
Hepatitis B virus genotype A1 subtype adw2 (isolate South Africa/84/2001) (HBV-A)
1 entry
Hepatitis B virus genotype A1 subtype adw2 (isolate Southern-Africa/Cai) (HBV-A)
1 entry
Hepatitis B virus genotype A2 (isolate Japan/11D11HCCW/1998) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw (isolate Japan/Nishioka/1983) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw2 (isolate Germany/991/1990) (HBV-A)
1 entry
Hepatitis B virus genotype A2 subtype adw2 (strain Rutter 1979) (HBV-A)
1 entry
Hepatitis B virus genotype A3 (isolate Cameroon/CMR711/1994) (HBV-A)
1 entry
Hepatitis B virus genotype A3 (isolate Cameroon/CMR983/1994) (HBV-A)
1 entry
Hepatitis B virus genotype B/C subtype adw (isolate Okinawa/pODW282/1998) (HBV-B)
1 entry
Hepatitis B virus genotype B1 (isolate Japan/Ry30/2002) (HBV-B)
1 entry
Hepatitis B virus genotype B1 (isolate Japan/Yamagata-2/1998) (HBV-B)
1 entry
Hepatitis B virus genotype B1 subtype adw (isolate Japan/pJDW233/1988) (HBV-B)
1 entry
Hepatitis B virus genotype B2 (isolate Indonesia/pIDW420/1988) (HBV-B)
1 entry
Hepatitis B virus genotype B2 (isolate Vietnam/16091/1992) (HBV-B)
1 entry
Hepatitis B virus genotype B2 (isolate Vietnam/9873/1997) (HBV-B)
1 entry
Hepatitis B virus genotype B2 subtype adw (isolate China/patient4/1996) (HBV-B)
1 entry
Hepatitis B virus genotype C (isolate Vietnam/3270/2000) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ad (isolate Japan/S-179/1988) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate China/NC-1/1988) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Japan/A4/1994) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Japan/Nishioka/1983) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (isolate Korea/Kim/1989) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype adr (strain Japan/adr4/1983) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ar (isolate Japan/S-207/1988) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ayw (isolate Australia/AustRC/1992) (HBV-C)
1 entry
Hepatitis B virus genotype C subtype ayw (isolate China/Tibet127/2002) (HBV-C)
1 entry
Hepatitis B virus genotype D (isolate France/alpha1/1989) (HBV-D)
1 entry
Hepatitis B virus genotype D (isolate Germany/1-91/1991) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype adw (isolate United Kingdom/adyw/1979) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Australia/AustKW/1991) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Italy/CI/1992) (HBV-D)
1 entry
Hepatitis B virus genotype D subtype ayw (isolate Japan/JYW796/1988) (HBV-D)
1 entry
Hepatitis B virus genotype E (isolate Cote d'Ivoire/ABI-129/2003) (HBV-E)
1 entry
Hepatitis B virus genotype E (isolate Cote d'Ivoire/ABI-212/2003) (HBV-E)
1 entry
Hepatitis B virus genotype E subtype ayw4 (isolate Kou) (HBV-E)
1 entry
Hepatitis B virus genotype F1 (isolate Argentina/sa11/2000) (HBV-F)
1 entry
Hepatitis B virus genotype F2 (isolate Argentina/sa16/2000) (HBV-F)
1 entry
Hepatitis B virus genotype F2 (isolate Brazil/w4B) (HBV-F)
1 entry
Hepatitis B virus genotype F2 subtype adw4q (isolate Senegal/9203) (HBV-F)
1 entry
Hepatitis B virus genotype G (isolate IG29227/2000) (HBV-G)
1 entry
Hepatitis B virus genotype H (isolate United States/LAS2523/2002) (HBV-H)
1 entry
Hepatitis B virus genotype H subtype adw4 (isolate Nicaragua/1853Nic/1997) (HBV-H)
1 entry
Hepatitis B virus genotype H subtype adw4 (isolate Nicaragua/2928Nic/1997) (HBV-H)
1 entry
Hepatitis C virus genotype 1a (isolate 1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate BK) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Con1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HC-J1) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HC-JT) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate HCR6) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Japanese) (HCV)
1 entry
Hepatitis C virus genotype 1b (isolate Taiwan) (HCV)
1 entry
Hepatitis C virus genotype 1b (strain HC-J4) (HCV)
1 entry
Hepatitis C virus genotype 1c (isolate HC-G9) (HCV)
1 entry
Hepatitis C virus genotype 1c (isolate India) (HCV)
1 entry
Hepatitis C virus genotype 2a (isolate HC-J6) (HCV)
1 entry
Hepatitis C virus genotype 2a (isolate JFH-1) (HCV)
1 entry
Hepatitis C virus genotype 2b (isolate HC-J8) (HCV)
1 entry
Hepatitis C virus genotype 2b (isolate JPUT971017) (HCV)
1 entry
Hepatitis C virus genotype 2c (isolate BEBE1) (HCV)
1 entry
Hepatitis C virus genotype 2k (isolate VAT96) (HCV)
1 entry
Hepatitis C virus genotype 3a (isolate NZL1) (HCV)
1 entry
Hepatitis C virus genotype 3a (isolate k3a) (HCV)
1 entry
Hepatitis C virus genotype 3b (isolate Tr-Kj) (HCV)
1 entry
Hepatitis C virus genotype 3k (isolate JK049) (HCV)
1 entry
Hepatitis C virus genotype 4a (isolate ED43) (HCV)
1 entry
Hepatitis C virus genotype 5a (isolate EUH1480) (HCV)
1 entry
Hepatitis C virus genotype 5a (isolate SA13) (HCV)
1 entry
Hepatitis C virus genotype 6a (isolate 6a33) (HCV)
1 entry
Hepatitis C virus genotype 6a (isolate EUHK2) (HCV)
1 entry
Hepatitis C virus genotype 6b (isolate Th580) (HCV)
1 entry
Hepatitis C virus genotype 6d (isolate VN235) (HCV)
1 entry
Hepatitis C virus genotype 6g (isolate JK046) (HCV)
1 entry
Hepatitis C virus genotype 6h (isolate VN004) (HCV)
1 entry
Hepatitis C virus genotype 6k (isolate VN405) (HCV)
1 entry
Human coronavirus HKU1 (isolate N2) (HCoV-HKU1)
1 entry
Human coronavirus HKU1 (isolate N5) (HCoV-HKU1)
1 entry
Human immunodeficiency virus type 1 group M subtype A (isolate MAL) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype A (isolate Z321) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate ARV2/SF2) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate BH10) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate BH8) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate BRU/LAI) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate BRVA) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate CDC-451) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate HXB3) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate JH32) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate JRCSF) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate KB-1/ETR) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate LW123) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate MFA) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate MN) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate NY5) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate OYI) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate RF/HAT3) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate SC) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate SF162) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate SF33) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate WMJ1) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate WMJ22) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (isolate YU-2) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype B (strain 89.6) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype C (isolate 92BR025) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype C (isolate ETH2220) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate ELI) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate NDK) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate Z2/CDC-Z34) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate Z6) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype D (isolate Z84) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype F1 (isolate 93BR020) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype F1 (isolate VI850) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype F2 (isolate MP255) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype F2 (isolate MP257) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype G (isolate 92NG083) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype H (isolate 90CF056) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype H (isolate VI991) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype J (isolate SE9173) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype J (isolate SE9280) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype K (isolate 96CM-MP535) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype K (isolate 97ZR-EQTB11) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group M subtype U (isolate Z3) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group N (isolate YBF106) (HIV-1)
1 entry
Human immunodeficiency virus type 1 group O (isolate MVP5180) (HIV-1)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate CAM2) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate D194) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate Ghana-1) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate KR) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate NIH-Z) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate ROD) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate SBLISY) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate ST) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype A (isolate ST/24.1C#2) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype B (isolate D205) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype B (isolate EHO) (HIV-2)
1 entry
Human immunodeficiency virus type 2 subtype B (isolate UC1) (HIV-2)
1 entry
Influenza A virus (strain A/Aichi/2/1968 H3N2)
1 entry
Influenza A virus (strain A/Beijing/353/1989 H3N2)
1 entry
Influenza A virus (strain A/Beijing/39/1975 H3N2)
1 entry
Influenza A virus (strain A/Brazil/11/1978 H1N1)
1 entry
Influenza A virus (strain A/Brevig Mission/1/1918 H1N1) (Influenza A virus (strain A/South Carolina/1/1918 H1N1))
1 entry
Influenza A virus (strain A/Budgerigar/Hokkaido/1/1977 H4N6)
1 entry
Influenza A virus (strain A/Chicken/Alabama/1/1975 H4N8)
1 entry
Influenza A virus (strain A/Chicken/Germany/n/1949 H10N7)
1 entry
Influenza A virus (strain A/Chicken/Hong Kong/220/1997 H5N1 genotype Gs/Gd)
1 entry
Influenza A virus (strain A/Chicken/Pennsylvania/1/1983 H5N2)
1 entry
Influenza A virus (strain A/Chicken/Scotland/1959 H5N1)
1 entry
Influenza A virus (strain A/Chicken/Victoria/1/1985 H7N7)
1 entry
Influenza A virus (strain A/Chicken/Weybridge H7N7) (Influenza A virus (strain A/FPV/Weybridge H7N7))
1 entry
Influenza A virus (strain A/Chile/1/1983 H1N1)
1 entry
Influenza A virus (strain A/China:Nanchang/11/1996 H1N1)
1 entry
Influenza A virus (strain A/Duck/Alberta/28/1976 H4N6)
1 entry
Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1)
1 entry
Influenza A virus (strain A/Duck/Alberta/60/1976 H12N5)
1 entry
Influenza A virus (strain A/Duck/Alberta/78/1976 H3N8)
1 entry
Influenza A virus (strain A/Duck/Australia/749/1980 H1N1)
1 entry
Influenza A virus (strain A/Duck/Czechoslovakia/1956 H4N6)
1 entry
Influenza A virus (strain A/Duck/England/1/1956 H11N6)
1 entry
Influenza A virus (strain A/Duck/Germany/1949 H10N7)
1 entry
Influenza A virus (strain A/Duck/Hokkaido/8/1980 H3N8)
1 entry
Influenza A virus (strain A/Duck/Hong Kong/2986.1/2000 H5N1 genotype C)
1 entry
Influenza A virus (strain A/Duck/Ireland/113/1983 H5N8)
1 entry
Influenza A virus (strain A/Duck/Memphis/928/1974 H3N8)
1 entry
Influenza A virus (strain A/Duck/New Zealand/31/1976 H4N6)
1 entry
Influenza A virus (strain A/Duck/Ukraine/1/1963 H3N8)
1 entry
Influenza A virus (strain A/England/321/1977 H3N2)
1 entry
Influenza A virus (strain A/Equine/Algiers/1972 H3N8)
1 entry
Influenza A virus (strain A/Equine/C.Detroit/1/1964 H7N7)
1 entry
Influenza A virus (strain A/Equine/Cambridge/1/1963 H7N7)
1 entry
Influenza A virus (strain A/Equine/Cambridge/1/1973 H7N7)
1 entry
Influenza A virus (strain A/Equine/Detroit/1/1964 H7N7)
1 entry
Influenza A virus (strain A/Equine/Fontainebleau/1976 H3N8) (Influenza A virus (strain A/Equine/France/1/1976 H3N8))
1 entry
Influenza A virus (strain A/Equine/Jillin/1/1989 H3N8)
1 entry
Influenza A virus (strain A/Equine/Kentucky/1/1981)
1 entry
Influenza A virus (strain A/Equine/Kentucky/1/1987 H3N8)
1 entry
Influenza A virus (strain A/Equine/Kentucky/2/1986 H3N8)
1 entry
Influenza A virus (strain A/Equine/Lexington/1/1966 H7N7)
1 entry
Influenza A virus (strain A/Equine/London/1416/1973 H7N7)
1 entry
Influenza A virus (strain A/Equine/Miami/1/1963 H3N8)
1 entry
Influenza A virus (strain A/Equine/New Market/1/1977 H7N7)
1 entry
Influenza A virus (strain A/Equine/New Market/1976 H3N8)
1 entry
Influenza A virus (strain A/Equine/Prague/1/1956 H7N7)
1 entry
Influenza A virus (strain A/Equine/Romania/1980 H3N8)
1 entry
Influenza A virus (strain A/Equine/Santiago/1/1985 H3N8)
1 entry
Influenza A virus (strain A/Equine/Sao Paulo/1/1976 H7N7)
1 entry
Influenza A virus (strain A/Equine/Suffolk/1989 H3N8)
1 entry
Influenza A virus (strain A/Equine/Switzerland/137/1972 H7N7)
1 entry
Influenza A virus (strain A/Equine/Tennessee/5/1986 H3N8)
1 entry
Influenza A virus (strain A/Equine/Tokyo/1971 H3N2)
1 entry
Influenza A virus (strain A/Equine/Uruguay/1/1963 H3N8)
1 entry
Influenza A virus (strain A/Fowl plague virus/Rostock/8/1934 H7N1)
1 entry
Influenza A virus (strain A/Grey teal/Australia/2/1979 H4N4)
1 entry
Influenza A virus (strain A/Gull/Astrakhan/227/1984 H13N6)
1 entry
Influenza A virus (strain A/Gull/Maryland/704/1977 H13N6)
1 entry
Influenza A virus (strain A/Gull/Minnesota/945/1980 H13N6)
1 entry
Influenza A virus (strain A/Henry/1936 H1N1)
1 entry
Influenza A virus (strain A/Herring gull/DE/677/1988 H2N8)
1 entry
Influenza A virus (strain A/Hickox/1940 H1N1)
1 entry
Influenza A virus (strain A/Hong Kong/1/1968 H3N2)
1 entry
Influenza A virus (strain A/Hong Kong/156/1997 H5N1 genotype Gs/Gd)
1 entry
Influenza A virus (strain A/Hong Kong/5/1983 H3N2)
1 entry
Influenza A virus (strain A/India/6263/1980 H1N1)
1 entry
Influenza A virus (strain A/Japan/305/1957 H2N2)
1 entry
Influenza A virus (strain A/Kiev/59/1979 H1N1)
1 entry
Influenza A virus (strain A/Korea/426/1968 H2N2)
1 entry
Influenza A virus (strain A/Leningrad/1/1954 H1N1)
1 entry
Influenza A virus (strain A/Malaysia:Malaya/302/1954 H1N1)
1 entry
Influenza A virus (strain A/Mallard/Astrakhan/263/1982 H14N5) (Influenza A virus (strain A/Mallard/Gurjev/263/1982 H14N5))
1 entry
Influenza A virus (strain A/Mallard/New York/6750/1978 H2N2)
1 entry
Influenza A virus (strain A/Mallard/New York/6874/1978 H3N2)
1 entry
Influenza A virus (strain A/Mallard/Ohio/556/1987 H5N9)
1 entry
Influenza A virus (strain A/Memphis/1/1971 H3N2)
1 entry
Influenza A virus (strain A/Memphis/101/1972 H3N2)
1 entry
Influenza A virus (strain A/Memphis/102/1972 H3N2)
1 entry
Influenza A virus (strain A/Memphis/110/1976 H3N2)
1 entry
Influenza A virus (strain A/Memphis/18/1978 H3N2)
1 entry
Influenza A virus (strain A/Memphis/2/1978 H3N2)
1 entry
Influenza A virus (strain A/Memphis/4/1980 H3N2)
1 entry
Influenza A virus (strain A/Memphis/6/1986 H3N2)
1 entry
Influenza A virus (strain A/New Zealand:South Canterbury/35/2000 H1N1)
1 entry
Influenza A virus (strain A/Northern Territory/60/1968 H3N2) (Influenza A virus (strain NT60)) (Influenza A virus (strain A/NT/60/1968 H3N2))
1 entry
Influenza A virus (strain A/Port Chalmers/1/1973 H3N2)
1 entry
Influenza A virus (strain A/Ruddy Turnstone/New Jersey/47/1985 H4N6)
1 entry
Influenza A virus (strain A/Russia:St.Petersburg/8/2006 H1N1)
1 entry
Influenza A virus (strain A/Seal/Massachusetts/1/1980 H7N7)
1 entry
Influenza A virus (strain A/Seal/Massachusetts/133/1982 H4N5)
1 entry
Influenza A virus (strain A/Shearwater/Australia/1972 H6N5)
1 entry
Influenza A virus (strain A/Singapore/1/1957 H2N2)
1 entry
Influenza A virus (strain A/Starling/Victoria/5156/1985 H7N7)
1 entry
Influenza A virus (strain A/Swine/Colorado/1/1977 H3N2)
1 entry
Influenza A virus (strain A/Swine/Indiana/1726/1988 H1N1)
1 entry
Influenza A virus (strain A/Swine/Iowa/15/1930 H1N1)
1 entry
Influenza A virus (strain A/Swine/Netherlands/12/1985 H1N1)
1 entry
Influenza A virus (strain A/Swine/New Jersey/11/1976 H1N1)
1 entry
Influenza A virus (strain A/Swine/Ukkel/1/1984 H3N2)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1961 H1N1)
1 entry
Influenza A virus (strain A/Swine/Wisconsin/1/1967 H1N1)
1 entry
Influenza A virus (strain A/Tern/South Africa/1961 H5N3)
1 entry
Influenza A virus (strain A/Turkey/Ireland/1378/1983 H5N8)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/1661/1981 H1N1)
1 entry
Influenza A virus (strain A/Turkey/Minnesota/833/1980 H4N2)
1 entry
Influenza A virus (strain A/Turkey/Ontario/6118/1968 H8N4)
1 entry
Influenza A virus (strain A/Turkey/Ontario/7732/1966 H5N9)
1 entry
Influenza A virus (strain A/Turkey/Oregon/1971 H7N3)
1 entry
Influenza A virus (strain A/Turkey/Wisconsin/1/1966 H9N2)
1 entry
Influenza A virus (strain A/USA:Albany/12/1951 H1N1)
1 entry
Influenza A virus (strain A/USA:Huston/AA/1945 H1N1)
1 entry
Influenza A virus (strain A/USA:Iowa/1943 H1N1)
1 entry
Influenza A virus (strain A/USA:Memphis/10/1996 H1N1)
1 entry
Influenza A virus (strain A/USA:Phila/1935 H1N1)
1 entry
Influenza A virus (strain A/USA:Texas/UR06-0195/2007 H1N1)
1 entry
Influenza A virus (strain A/USSR/90/1977 H1N1)
1 entry
Influenza A virus (strain A/Udorn/307/1972 H3N2)
1 entry
Influenza A virus (strain A/Victoria/3/1975 H3N2)
1 entry
Influenza A virus (strain A/Whale/Maine/328/1984 H13N2)
1 entry
Influenza A virus (strain A/Wilson-Smith/1933 H1N1) (Influenza A virus (strain A/WS/1933 H1N1))
1 entry
Influenza A virus (strain A/X-31 H3N2)
1 entry
Influenza B virus (strain B/Beijing/1/1987)
1 entry
Influenza B virus (strain B/England/222/1982)
1 entry
Influenza B virus (strain B/Oregon/5/1980)
1 entry
Influenza B virus (strain B/Singapore/222/1979)
1 entry
Influenza B virus (strain B/Victoria/2/1987)
1 entry
Influenza C virus (strain C/California/1978)
1 entry
Influenza C virus (strain C/Johannesburg/1/1966)
1 entry
Influenza C virus (strain C/Nara/2/1985)
1 entry
Influenza C virus (strain C/Yamagata/4/1988)
1 entry
Junin mammarenavirus (JUNV) (Junn mammarenavirus)
1 entry
Kunjin virus (strain MRM61C)
1 entry
Lake Victoria marburgvirus (strain Angola/2005) (MARV)
1 entry
Lake Victoria marburgvirus (strain Ozolin-75) (MARV) (Marburg virus (strain South Africa/Ozolin/1975))
1 entry
Lake Victoria marburgvirus (strain Popp-67) (MARV) (Marburg virus (strain West Germany/Popp/1967))
1 entry
Lake Victoria marburgvirus (strain Ravn-87) (MARV) (Marburg virus (strain Kenya/Ravn/1987))
1 entry
Langat virus (strain TP21)
1 entry
Lassa virus (strain GA391) (LASV)
1 entry
Louping ill virus (isolate Grouses/Scotland/K/1980) (LIV)
1 entry
Lymphocytic choriomeningitis virus (strain WE) (LCMV)
1 entry
Machupo virus (MACV)
1 entry
Middelburg virus
1 entry
Middle East respiratory syndrome-related coronavirus (isolate United Kingdom/H123990006/2012) (MERS-CoV) (Betacoronavirus England 1)
1 entry
Mopeia virus (MOPV)
1 entry
Murine coronavirus (strain 4) (MHV-4) (Murine hepatitis virus)
1 entry
Murine coronavirus (strain JHM) (MHV-JHM) (Murine hepatitis virus)
1 entry
Murine coronavirus (strain JHMV / variant CL-2) (MHV) (Murine hepatitis virus)
1 entry
New York virus (NYV)
1 entry
O'nyong-nyong virus (strain Gulu) (ONNV)
1 entry
O'nyong-nyong virus (strain Igbo Ora) (ONNV) (Igbo Ora virus)
1 entry
Orangutan hepatitis B virus (isolate Somad) (HBVoru)
1 entry
Porcine hemagglutinating encephalomyelitis virus (strain 67N) (HEV-67N)
1 entry
Porcine hemagglutinating encephalomyelitis virus (strain IAF-404) (HEV)
1 entry
Punta toro phlebovirus
1 entry
Puumala virus (strain K27)
1 entry
Puumala virus (strain P360)
1 entry
Puumala virus (strain Sotkamo/V-2969/81)
1 entry
Rat coronavirus (strain 681) (RCV-SDAV) (Sialodacryoadenitis virus SDAV-681)
1 entry
Reston ebolavirus (strain Philippines-96) (REBOV) (Reston Ebola virus)
1 entry
Reston ebolavirus (strain Siena/Philippine-92) (REBOV) (Reston Ebola virus)
1 entry
Rift valley fever virus (RVFV)
1 entry
Rocio virus (ROCV)
1 entry
Ross river virus (strain NB5092) (RRV)
1 entry
Ross river virus (strain T48) (RRV)
1 entry
Rubella virus (strain BRD1) (RUBV)
1 entry
Rubella virus (strain BRDII) (RUBV)
1 entry
Rubella virus (strain Cendehill) (RUBV)
1 entry
Rubella virus (strain M33) (RUBV)
1 entry
Rubella virus (strain RA27/3 vaccine) (RUBV)
1 entry
Rubella virus (strain RN-UK86) (RUBV)
1 entry
Rubella virus (strain TO-336 vaccine) (RUBV)
1 entry
Rubella virus (strain TO-336) (RUBV)
1 entry
Sagiyama virus (SAGV)
1 entry
Seoul virus (strain R22)
1 entry
Seoul virus (strain SR-11) (Sapporo rat virus)
1 entry
Simian immunodeficiency virus (isolate CPZ GAB1) (SIV-cpz) (Chimpanzee immunodeficiency virus)
1 entry
Simian immunodeficiency virus (isolate EK505) (SIV-cpz) (Chimpanzee immunodeficiency virus)
1 entry
Simian immunodeficiency virus (isolate MB66) (SIV-cpz) (Chimpanzee immunodeficiency virus)
1 entry
Sindbis virus subtype Ockelbo (strain Edsbyn 82-5) (OCKV) (Ockelbo virus)
1 entry
Sudan ebolavirus (strain Boniface-76) (SEBOV) (Sudan Ebola virus)
1 entry
Sudan ebolavirus (strain Maleo-79) (SEBOV) (Sudan Ebola virus)
1 entry
Tacaribe virus (strain TRVL 11598) (TCRV)
1 entry
Tacaribe virus (strain V5) (TCRV)
1 entry
Tacaribe virus (strain V7) (TCRV)
1 entry
Tai Forest ebolavirus (strain Cote d'Ivoire-94) (TAFV) (Cote d'Ivoire Ebola virus)
1 entry
Tick-borne encephalitis virus (strain Hypr) (TBEV)
1 entry
Tick-borne encephalitis virus Far Eastern subtype (strain Sofjin) (SOFV) (Sofjin virus)
1 entry
Tomato spotted wilt virus (TSWV)
1 entry
Tomato spotted wilt virus (isolate D) (TSWV)
1 entry
Tomato spotted wilt virus (strain Regular2A) (TSWV)
1 entry
Venezuelan equine encephalitis virus (strain 3880) (VEEV)
1 entry
Venezuelan equine encephalitis virus (strain Everglades Fe3-7c) (VEEV)
1 entry
Venezuelan equine encephalitis virus (strain Mena II) (VEEV)
1 entry
Venezuelan equine encephalitis virus (strain P676) (VEEV)
1 entry
Venezuelan equine encephalitis virus (strain TC-83) (VEEV)
1 entry
Vesicular stomatitis Indiana virus (strain 85CLB South America) (VSIV)
1 entry
Vesicular stomatitis Indiana virus (strain 94GUB Central America) (VSIV)
1 entry
Vesicular stomatitis Indiana virus (strain Glasgow) (VSIV)
1 entry
Vesicular stomatitis Indiana virus (strain Mudd-Summers) (VSIV)
1 entry
Vesicular stomatitis Indiana virus (strain Orsay) (VSIV)
1 entry
Vesicular stomatitis New Jersey virus (strain Ogden subtype Concan) (VSNJV)
1 entry
West Nile virus (strain NY-99) (WNV) (West Nile virus (strain NY-1999))
1 entry
Western equine encephalitis virus (WEEV)
1 entry
Woodchuck hepatitis B virus (isolate 1) (WHV)
1 entry
Woodchuck hepatitis B virus (isolate 2) (WHV)
1 entry
Woodchuck hepatitis B virus (isolate 59) (WHV)
1 entry
Woodchuck hepatitis B virus (isolate 8) (WHV)
1 entry
Yellow fever virus (isolate Angola/14FA/1971) (YFV)
1 entry
Yellow fever virus (isolate Ethiopia/Couma/1961) (YFV)
1 entry
Yellow fever virus (isolate Ivory Coast/1999) (YFV)
1 entry
Yellow fever virus (isolate Ivory Coast/85-82H/1982) (YFV)
1 entry
Yellow fever virus (isolate Uganda/A7094A4/1948) (YFV)
1 entry
Yellow fever virus (strain French neurotropic vaccine FNV) (YFV)
1 entry
Yellow fever virus (strain Ghana/Asibi/1927) (YFV)
1 entry
Yellow fever virus (strain Trinidad/TRINID79A/1979) (YFV)
1 entry
Zaire ebolavirus (strain Eckron-76) (ZEBOV) (Zaire Ebola virus)
1 entry
Zaire ebolavirus (strain Gabon-94) (ZEBOV) (Zaire Ebola virus)
1 entry
Zaire ebolavirus (strain Kikwit-95) (ZEBOV) (Zaire Ebola virus)
1 entry
Zika virus (isolate ZIKV/Human/Cambodia/FSS13025/2010) (ZIKV)
1 entry
